Your selection
Research, Patient care / 17.04.2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect
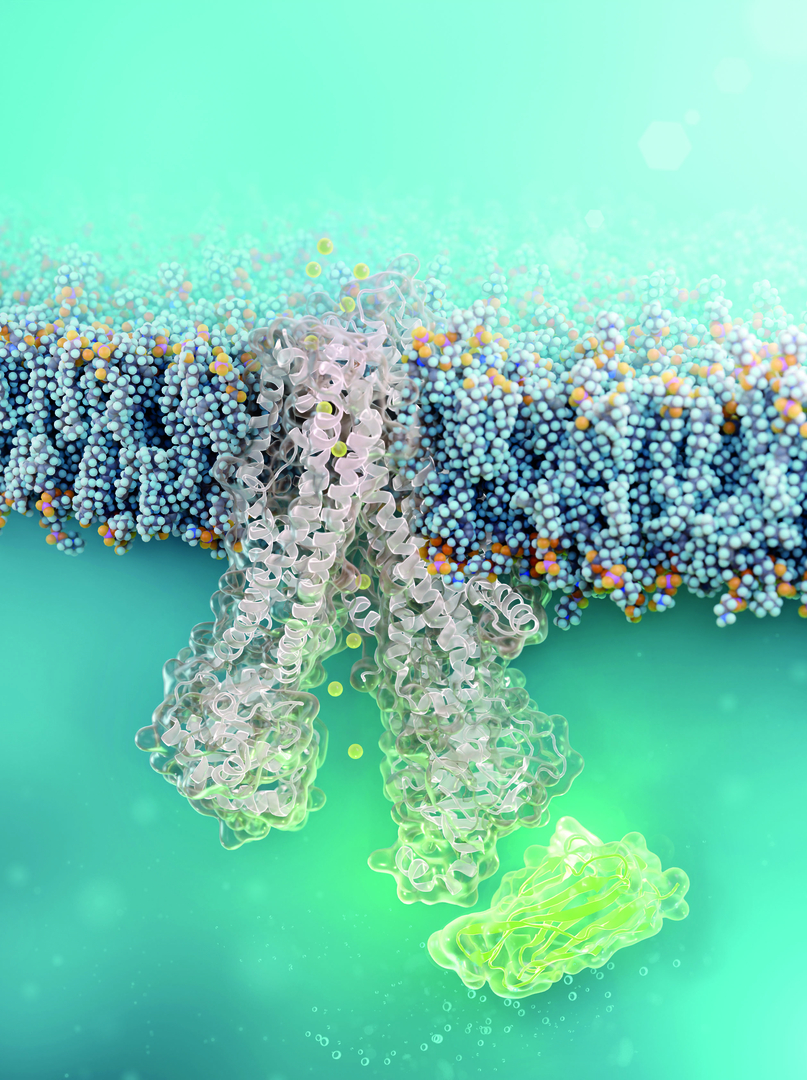
A tiny antibody component could fundamentally transform the treatment of cystic fibrosis: For the first time, researchers have succeeded in developing a so-called nanobody that penetrates directly into human cells and can repair the chloride channel most commonly affected in cystic fibrosis. The innovative therapeutic approach was developed in collaboration between teams from the Leibniz-Forschungsinstitut für Molekulare Pharmakologie (FMP) and Charité – Universitätsmedizin Berlin.The first authors of the study, which was recently published in the renowned journal Nature Chemical Biology, are Luise Franz (FMP) and Tihomir Rubil (Charité).
The clinical picture of cystic fibrosis—also known as CF—is caused by genetic defects in the so-called CFTR channel. This channel regulates water and salt transport in the lung mucosa and ensures the production of sufficiently fluid mucus.
In about 90 percent of cystic fibrosis patients, a mutation known as F580del is present in the CFTR channel, meaning that a single amino acid is missing at position 508 in its protein chain. This change causes CFTR to fold incorrectly and be broken down prematurely inside the cell, rather than functioning as a channel in the cell membrane of the airways. As a result, patients have thick mucus in their lungs, and pathogens can no longer be effectively cleared. The consequence is chronic infection and inflammation of the airways, leading to a progressive loss of lung function—in the worst-case scenario, this necessitates a lung transplant.
Professor Dr. Marcus Mall, Director of the Department of Pediatric Respiratory Medicine, Immunology and Critical Care Medicine at Charité, has, together with his team, made a significant contribution in recent years to noticeably improving the treatment of cystic fibrosis through therapy with three small-molecule drugs (CFTR modulators): With the help of the so-called triple therapy consisting of elexacaftor, tezacaftor, and ivacaftor (ETI), the function of the CFTR channel can be increased to about 50 percent of the normal level. However, chronic inflammation and infection of the lungs often persist, and there are also patients for whom this therapy is ineffective or whom cannot tolerate it.
An Antibody as a Repair Aid
There may be additional treatment options for this group in the future: The team led by chemist Professor Dr. Christian Hackenberger at the Leibniz-FMP has developed a new molecule in the lab that stabilizes the misfolded CFTR directly inside the cell. This is a nanobody—a tiny but stable antibody component that can bind precisely to defined surfaces of proteins. It is chemically modified with a “transport signal,” known as cell-penetrating peptides, which help it penetrate directly into the lung’s mucosal cells. There, the nanobody binds to the defective channel protein and helps it adopt the correct shape.
The researchers were able to demonstrate that the nanobody remained bound to the mutated CFTR channel in cells derived from cystic fibrosis patients for at least 24 hours. It did not damage the cells in the process. Functional studies also confirmed that the corrected channel once again transported chloride across the cell membrane.
Combination of triple therapy and nanobody
In combination with established ETI triple therapy, the nanobody demonstrates a pronounced synergistic effect in these cell cultures: While the ETI agents restored the function of the defective CFTR channel by about half on average, the channel activity could be increased to nearly 90 percent of normal levels through the additional administration of the nanobody.
The study thus demonstrates that exogenously administered cell-penetrating nanobodies can stabilize disease-relevant, misfolded proteins inside cells and restore their function. “In addition to the preclinical proof of concept for repairing the CFTR channel, this is the first example of a functional cell-permeable antibody: Until now, cell-permeable nanobodies have primarily been used to visualize intracellular target structures or for the targeted killing of cells,” says Prof. Dr. Christian Hackenberger.
“Since the nanobodies bind directly in the region of the F508del mutation, they enable even more targeted treatment of the maturation defect in CFTR channels,” says Prof. Dr. Marcus Mall. “This new mechanism of action allows CFTR function to be corrected significantly better in combination with existing CFTR modulators. Our results suggest that this new approach may even enable complete normalization of CFTR function. This would be another breakthrough for the treatment of cystic fibrosis.”
Thus, this work thus opens up new possibilities for further improving the treatment of cystic fibrosis—while also laying the groundwork for broader therapeutic applications.
Prospects beyond cystic fibrosis
However, key questions must still be resolved before the approach can be applied clinically to cystic fibrosis, such as developing a suitable formulation for inhalation and ensuring efficient penetration of the viscous CF mucus. Furthermore, it remains unclear how the nanobody acts within the body and how the immune system reacts to nanobody treatment. These challenges are currently being addressed within Collaborative Research Center 1449 “Dynamic Hydrogels at Biointerfaces,” within the framework of which the current results were also generated.
The approach of intracellular nanobody therapy could also be helpful beyond cystic fibrosis for other rare genetic diseases in which protein misfolding plays a role and for which there are currently few effective treatments.
About Cystic Fibrosis
Cystic fibrosis is one of the most common fatal hereditary diseases worldwide. As many as 8,000 children, teens, and adults are living with the disease in Germany today. An imbalance in salt and water levels in the body causes people with cystic fibrosis to produce thick, sticky secretions that harm organs such as the lungs and pancreas. This leads to progressive loss of lung function and shortness of breath, which still significantly lowers life expectancy despite advances in treatment. Some 150 to 200 children are born with this rare disease in Germany each year. A test for cystic fibrosis is part of routine screening for newborns.
Publication: Franz, L., Rubil, T., Balázs, A., Overtus, M., Kemnitz-Hassanin, K., Govaerts, C., Mall, M. A., & Hackenberger, C. P. R.. A cell-permeable nanobody to restore F508del cystic fibrosis transmembrane conductance regulator activity. Nature Chemical Biology 2026. doi: 10.1038/s41589-026-02199-w
Source: Joint Press Release from the Leibniz-Forschungsinstitut für Molekulare Pharmakologie and Charité – Universitätsmedizin Berlin
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect
Overview News
News Buch Berlin
First Joint Career Day Brings Science and Industry Together
The Max Delbrück Center’s Career Day, held in cooperation with the operator of the Berlin-Buch Campus, offered young researchers insights into the working world of startups and biotech companies
more ...A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect
A tiny antibody component could fundamentally transform the treatment of cystic fibrosis: For the first time, researchers have succeeded in developing a so-called nanobody that penetrates directly int...
more ...Eckert & Ziegler: Metzler Initiates Coverage with a Buy Rating and a Price Target of € 21.00. Upside Potential: 41%
Eckert & Ziegler: Metzler Initiates Coverage with a Buy Rating and a Price Target of € 21.00. Upside Potential: 41%
more ...Events Buch Berlin
08.05.2026, 11:00
Kundgebung am 8. Mai "Nie wieder Krieg, nie wieder Faschismus"
Das Netzwerk gegen Rassismus Buch-Karow, die Linke, B90/Grüne, VVN-BdA u.a. rufen am Freitag zur Kundgebung "Nie wieder Krieg, nie wieder Faschismus" auf.
more ...08.05.2026, 14:00
Aktionstag "Offenes Buch - offene Türen"
Der Stadtteil Buch kann am Freitag, 8. Mai, bei der Veranstaltung "Offenes Buch - offene Türen" entdeckt werden. Soziale und kulturelle Angebot für Kinder, Jugendliche, Familien und Senioren gibt es d...
more ...08.05.2026, 16:00
Dialog an Deck: Chancen und Risiken von Gen- und Zelltherapien
Im Rahmen des Wissenschaftsjahres 2026 „Medizin der Zukunft“ lädt das Berlin Institute of Health in der Charité (BIH) zu einer Podiumsdiskussion auf der MS Wissenschaft ein. Expert*innen aus Forschung...
more ...






