News
Research, Innovation, Patient care / 22.05.2026
Berlin honors outstanding deep-tech companies—including MyoPax

This year, for the first time, a Deep Tech Award was presented in the Bio & Health Tech category. The winner is MyoPax, a spin-off of the Max Delbrück Center and Charité–Universitätsmedizin Berlin.
AI-powered fact-checking to combat disinformation, novel cell therapies for muscle tissue regeneration, sustainable 3D printing, and decentralized satellite communications: On the evening of Wednesday, May 20, 2026, the Senate Department for Economic Affairs, Energy and Public Enterprises presented the Deep Tech Award to five outstanding Berlin-based companies. For the eleventh consecutive year, the administration is thus recognizing technological, research-based innovations from Berlin that stand out for their practical application, societal relevance, and added value. The awards ceremony, attended by Senator for Economic Affairs Franziska Giffey and Permanent Secretary for Economic Affairs Michael Biel, took place for the first time as part of the Deep Tech Momentum conference at Wilhelm Studios in Berlin.
Franziska Giffey, Mayor and Senator for Economic Affairs, Energy and Public Enterprises: “We want to make Berlin the number one hub for innovation in Europe. The companies honored today are turning cutting-edge technological research into practical applications and offering solutions to current challenges. In doing so, they impressively demonstrate the immense innovative potential that Berlin possesses. With the Deep Tech Award, we recognize the courage and excellence of our founders. Through our new partnership with the Deep Tech Momentum conference, we are also creating a platform that connects Berlin’s brightest minds even more closely with industry partners and international investors. In doing so, we are ensuring sustainable growth and technological sovereignty for Berlin as a hub.”
The Deep Tech Award offers a total prize pool of 50,000 euros and is presented in five categories: “Advanced Manufacturing,” “Bio- & Healthtech,” “Artificial Intelligence,” “Quantum Technologies, Photonics & Microelectronics,” and “Web3 & Distributed Ledger Technologies (DLT).” From over 84 applications, expert juries selected five winning companies, each of which will receive prize money of 10,000 euros.
With the Deep Tech Award being integrated into Deep Tech Momentum for the first time, the prize is being positioned even more strongly on the international stage. The conference is considered one of Europe’s leading platforms for connecting deep-tech startups with companies and investors. The new partnership underscores the award’s mission to not only honor Berlin’s most innovative technology companies but also to connect them even more closely with the European innovation ecosystem. In this context, the special “Deep Tech Award for Breakthrough Momentum” was also presented for the first time this year. It is aimed at European startups curated by Deep Tech Momentum that combine excellent scientific innovation with exceptional scaling potential.
The winners of the 2026 Deep Tech Award at a glance:
Deep Tech Star in the “Advanced Manufacturing” category: Endless Industries GmbH
Endless Industries is revolutionizing the manufacturing of fiber-reinforced composites—materials in which carbon fibers are embedded in a binding resin—with a 3D printing solution. This makes it possible to replace complex and expensive manufacturing processes and reduce waste. Website:
www.endless.industries
Deep Tech Star in the “Bio- & Healthtech” category: MyoPax GmbH
MyoPax’s work focuses on innovative cell therapies and gene corrections for the regeneration of muscle tissue. In doing so, the company specifically targets severe muscle injuries and diseases, offering patients promising new treatment options. Website
: www.myopax.com
Deep Tech Star in the “Artificial Intelligence” category: Gretchen AI GmbH
Gretchen AI develops state-of-the-art AI to detect deepfakes and fake news and can reconstruct their dissemination history. This enables the Berlin-based company to help major media organizations conduct fact-checks up to six times faster while maintaining the same level of reliability—a crucial contribution to safeguarding the public information space.
Website: www.gretchen-ai.com
Deep Tech Star in the “Quantum Technologies, Photonics & Microelectronics” category: Xavveo GmbH
Xavveo develops photonic radar sensors that set new standards in fields such as navigation and measurement technology, for example in environmental sensing for automobiles. The technology enables unprecedented precision and has the potential to fundamentally replace existing sensor solutions across a wide range of industries.
Website: www.xavveo.com
Deep Tech Star in the “Web3 & Distributed Ledger Technologies (DLT)” category: Decen Space UG
The startup Decen Space is developing a decentralized coordination network consisting of software and hardware components for the secure and efficient synchronization of data streams between satellites and ground stations. This solution enables higher data transfer rates at significantly lower costs and also allows satellite operators more contact time with their satellites. Website
: www.decenspace.com
Deep Tech Star of the Special Prize “Deep Tech Award for Breakthrough Momentum”: Six Robotics AS
Six Robotics AS is a Norwegian company specializing in the development of autonomy software for fleets of unmanned aerial vehicles. The software is based on innovative swarm intelligence algorithms and real-time mission control architectures that ensure a high degree of autonomy and efficiency for the aircraft. This enables the company’s drones to coordinate and execute missions as intelligent teams. As a result, the use of networked autonomous systems in modern defense operations is being advanced. Website: www.sixrobotics.com
For more information about Deep Tech Berlin and the Deep Tech Award, please visit: www.berlin.de/deeptech/
Source: Press release of the Senate Department for Economic Affairs, Energy and Public Enterprises
Innovation / 22.05.2026
Eckert & Ziegler Receives “Best Managed Companies Award” Once Again
Eckert & Ziegler SE (ISIN DE0005659700) has won the “Best Managed Companies Award” for the third time in a row. With this honor, Deloitte Private, UBS, Frankfurter Allgemeine Zeitung, and the Federation of German Industries (BDI) recognize excellently managed medium-sized companies.
“Good corporate leadership is of central importance, especially in economically challenging times. The Best Managed Companies Award is a well‑deserved recognition for companies that successfully combine responsibility, foresight, and sustainable action,” says Tobias Vogel, CEO of UBS Europe SE.
“This award is both recognition and motivation for us. It confirms that we are on the right path with a clear strategy in the growing nuclear medicine market—together with a fantastic team around the world,” added Dr Dirk W. Becker, a member of the Group Executive Committee of Eckert & Ziegler SE, who accepted the corporate trophy on behalf of the company at the awards ceremony in Frankfurt am Main.
The award is the result of a comprehensive, multi-stage application process in which companies are assessed for their excellence in the core areas of strategy, productivity and innovation, culture and commitment as well as finance and governance. A consistently high level of performance in all four categories is a prerequisite for selection. The final decision is made by an independent jury made up of renowned experts from business, science and the media.
About Eckert & Ziegler.
Eckert & Ziegler SE with more than 1,000 employees, is a leading specialist for isotope-related components in nuclear medicine and radiation therapy. The company offers a broad range of services and products for the radiopharmaceutical industry, from early development work to contract manufacturing and distribution. Eckert & Ziegler shares (ISIN DE0005659700) are listed in the TecDAX index of Deutsche Börse.
Contributing to saving lives.
Innovation / 12.05.2026
Eckert & Ziegler with a Successful Start to the Year. 2026 Forecast Confirmed.
1st Quarter 2026:
- Sales of €72.9 million (previous year: €68.2 million)
- EBIT before special items of €16.0 million (previous year: €16.2 million)
- Net income of €10.4 Mio. (previous year: €9.7 million)
Forecast 2026:
- Sales of around €320 million (confirmed)
- EBIT before special items of around €80 million (confirmed)
Eckert & Ziegler SE (ISIN DE0005659700, TecDAX) increased its sales in the first quarter of 2026 by 7% to €72.9 million compared with the same period last year. Due to a slightly weaker product mix in the Isotope Products segment during the first two months of the year, adjusted Group EBIT decreased by 2% to €16.0 million. Net income increased by 7% to €10.4 million, or €0.17 per share.
Sales in the Medical segment were significantly higher in the first three months of the year at €41.5 million compared to the previous year (€34.4 million). The pharmaceutical radioisotope business remains the most important revenue driver. In particular, the development of sales of generators and in the Contract Manufacturing & Development (CDMO) segment is worth noting.
The Isotope Products segment generated sales of €31.5 million, which was €2.3 million, or approximately 7%, lower than in the first three months of the previous year. A strong fourth quarter of 2025 was followed by a subdued start to the year, which regained significant momentum in March.
The forecast for fiscal year 2026, published on 26 March 2026, remains unchanged. The Executive Board continues to anticipate revenue of approximately €320 million and adjusted EBIT of approximately €80 million.
The complete quarterly report can be viewed here: https://www.ezag.com/Q12026en
About Eckert & Ziegler.
Eckert & Ziegler SE with more than 1,000 employees is a leading specialist for isotope-related components in nuclear medicine and radiation therapy. The company offers a broad range of services and products for the radiopharmaceutical industry, from early development work to contract manufacturing and distribution. Eckert & Ziegler shares (ISIN DE0005659700) are listed in the TecDAX index of Deutsche Börse.
Contributing to saving lives.
Research, Innovation, Patient care, Education / 22.04.2026
First Joint Career Day Brings Science and Industry Together

The Max Delbrück Center’s Career Day, held in cooperation with the operator of the Berlin-Buch Campus, offered young researchers insights into the working world of startups and biotech companies
April 16 was a packed day for participants in the Max Delbrück Center’s Career Day, titled “Transition Possible – Explore Careers Beyond R&D.” Numerous doctoral students and postdocs gained insights into potential careers outside the academic world.
The event was opened by biologist Lars Dittrich, who works as a science editor at MaiThink X. In the morning, there were virtual presentations across the Helmholtz Association on career paths outside of research. The speakers presented concrete entry-level and development opportunities and shared their experiences of transitioning from science to other professional fields.
In the afternoon, the Max Delbrück Center and Campus Berlin-Buch GmbH hosted an event at the BerlinBioCube startup center. Here, the Berlin BioScience Academy and the Innovation & Entrepreneurship Department of the Max Delbrück Center presented themselves. This was followed by lab tours at biotech companies and a workshop on the application process.
Pathways into the Biotech and Pharmaceutical Industries
How can you gain in-depth insights into the biotech and pharmaceutical industries without actually working in them? The Berlin BioScience Academy (BBA) offers exactly this opportunity. Its courses cover biotechnological and pharmacological processes, including Good Manufacturing Practice (GMP) and Good Clinical Practice (GCP). “Anyone considering a move into the industry or wanting to take the leap into a startup will gain an overview of the entire drug development process—from concept to market—in a very short time at the Biotech & Pharma Summer School,” said Dr. Uwe Lohmeier, who heads the BBA. Through its “Talk im Cube” event series, the BBA regularly brings together science and industry, offering panel discussions on topics such as financing strategies, female founders, CROs, and IP strategies in biotechnology. Here, too, participants have an easy opportunity to engage with biotech companies.
At the Max Delbrück Center, the Innovation & Entrepreneurship Department serves as a springboard for careers in spin-offs. Dr. Nevine Shalaby highlighted funding opportunities for future “sciencepreneurs” to develop innovative diagnostic or therapeutic approaches for practical application. The Innovation Office provides support through programs such as BOOST and PreGoBio to validate ideas and their basic feasibility, helps secure funding, offers mentoring, establishes contacts with industry and investors, and proactively supports business development.
How do biotech companies operate?
Over 50 biotech and medtech companies have set up their business in the BiotechPark Berlin-Buch, including numerous startups. Four of them opened their doors on Career Day to show participants how they work and what their mission is. T-knife, a spin-off of the Max Delbrück Center and Charité, introduced itself as a young biopharmaceutical company developing next-generation T-cell therapies to fight cancer.
CheckImmune, a spin-off of Charité, provided information about its work as an accredited specialized laboratory that supports the clinical development of new therapeutics through immunological studies, among other activities.
In the Biosynth laboratories, participants learned about the technologies used to develop and manufacture polymer-based excipients for drug delivery as well as bioconjugate drugs. Last but not least, the FyoniBio team presented its range of contract development and clinical laboratory services.
“There are surprisingly many different companies here,” said one participant, who could well imagine working at one of the biotech firms: “The labs in the BioCube are similar to those at research institutions, and the building feels very spacious, especially thanks to the large shared large common areas with a kitchen.” One of the participants noted with pleasant surprise how diverse the age range of employees in startups can be. Many found it fascinating to learn how the work culture in a startup functions and that the tasks there are different and more varied than in pure scientific research.
How successful is my application?
To wrap up the event, Career Day offered participants the chance to step into the shoes of a hiring manager during a group workshop. Anita Überheim, Head of Human Resources Europe at the global company Eckert & Ziegler SE, had the participants evaluate three anonymized CVs and cover letters and then explained which aspects matter in the selection process. She described how HR professionals proceed, how much time they have to review applications, which skills are important to mention, and what mistakes are common. Finally, the expert conducted a brief mock interview with one of the participants. During the joint evaluation with the audience, she explained how applicants should best communicate and respond. In addition to many helpful tips, a key insight for the young talents was this: It is not always necessary to meet 90 percent of the desired qualifications. What is far more important is that the person fits into the team and has the potential to continue developing.
The joint Career Day was very well received. “We are delighted to have had the opportunity to help organize this event. The Career Day offers concrete insights into the biotech industry and connects young talent with potential employers in the area, which is valuable for everyone involved,” says Dr. Ulrich Scheller, Managing Director of Campus Berlin-Buch GmbH.
Research, Patient care / 17.04.2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect
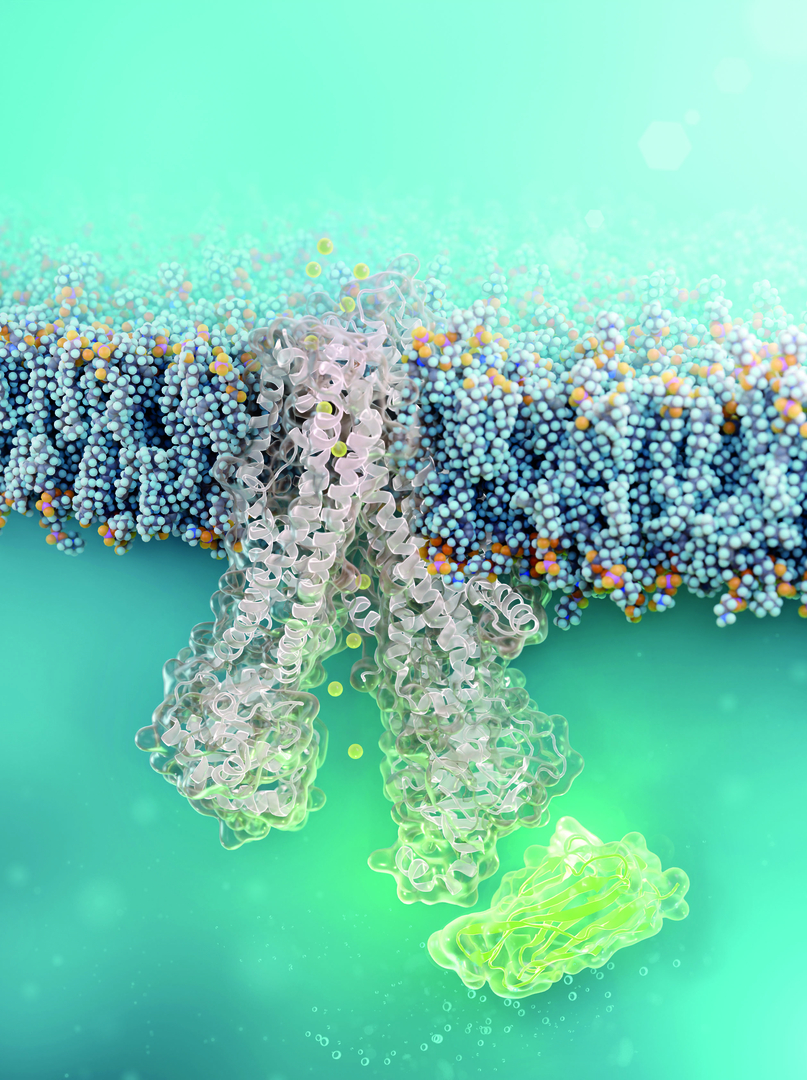
A tiny antibody component could fundamentally transform the treatment of cystic fibrosis: For the first time, researchers have succeeded in developing a so-called nanobody that penetrates directly into human cells and can repair the chloride channel most commonly affected in cystic fibrosis. The innovative therapeutic approach was developed in collaboration between teams from the Leibniz-Forschungsinstitut für Molekulare Pharmakologie (FMP) and Charité – Universitätsmedizin Berlin.The first authors of the study, which was recently published in the renowned journal Nature Chemical Biology, are Luise Franz (FMP) and Tihomir Rubil (Charité).
The clinical picture of cystic fibrosis—also known as CF—is caused by genetic defects in the so-called CFTR channel. This channel regulates water and salt transport in the lung mucosa and ensures the production of sufficiently fluid mucus.
In about 90 percent of cystic fibrosis patients, a mutation known as F580del is present in the CFTR channel, meaning that a single amino acid is missing at position 508 in its protein chain. This change causes CFTR to fold incorrectly and be broken down prematurely inside the cell, rather than functioning as a channel in the cell membrane of the airways. As a result, patients have thick mucus in their lungs, and pathogens can no longer be effectively cleared. The consequence is chronic infection and inflammation of the airways, leading to a progressive loss of lung function—in the worst-case scenario, this necessitates a lung transplant.
Professor Dr. Marcus Mall, Director of the Department of Pediatric Respiratory Medicine, Immunology and Critical Care Medicine at Charité, has, together with his team, made a significant contribution in recent years to noticeably improving the treatment of cystic fibrosis through therapy with three small-molecule drugs (CFTR modulators): With the help of the so-called triple therapy consisting of elexacaftor, tezacaftor, and ivacaftor (ETI), the function of the CFTR channel can be increased to about 50 percent of the normal level. However, chronic inflammation and infection of the lungs often persist, and there are also patients for whom this therapy is ineffective or whom cannot tolerate it.
An Antibody as a Repair Aid
There may be additional treatment options for this group in the future: The team led by chemist Professor Dr. Christian Hackenberger at the Leibniz-FMP has developed a new molecule in the lab that stabilizes the misfolded CFTR directly inside the cell. This is a nanobody—a tiny but stable antibody component that can bind precisely to defined surfaces of proteins. It is chemically modified with a “transport signal,” known as cell-penetrating peptides, which help it penetrate directly into the lung’s mucosal cells. There, the nanobody binds to the defective channel protein and helps it adopt the correct shape.
The researchers were able to demonstrate that the nanobody remained bound to the mutated CFTR channel in cells derived from cystic fibrosis patients for at least 24 hours. It did not damage the cells in the process. Functional studies also confirmed that the corrected channel once again transported chloride across the cell membrane.
Combination of triple therapy and nanobody
In combination with established ETI triple therapy, the nanobody demonstrates a pronounced synergistic effect in these cell cultures: While the ETI agents restored the function of the defective CFTR channel by about half on average, the channel activity could be increased to nearly 90 percent of normal levels through the additional administration of the nanobody.
The study thus demonstrates that exogenously administered cell-penetrating nanobodies can stabilize disease-relevant, misfolded proteins inside cells and restore their function. “In addition to the preclinical proof of concept for repairing the CFTR channel, this is the first example of a functional cell-permeable antibody: Until now, cell-permeable nanobodies have primarily been used to visualize intracellular target structures or for the targeted killing of cells,” says Prof. Dr. Christian Hackenberger.
“Since the nanobodies bind directly in the region of the F508del mutation, they enable even more targeted treatment of the maturation defect in CFTR channels,” says Prof. Dr. Marcus Mall. “This new mechanism of action allows CFTR function to be corrected significantly better in combination with existing CFTR modulators. Our results suggest that this new approach may even enable complete normalization of CFTR function. This would be another breakthrough for the treatment of cystic fibrosis.”
Thus, this work thus opens up new possibilities for further improving the treatment of cystic fibrosis—while also laying the groundwork for broader therapeutic applications.
Prospects beyond cystic fibrosis
However, key questions must still be resolved before the approach can be applied clinically to cystic fibrosis, such as developing a suitable formulation for inhalation and ensuring efficient penetration of the viscous CF mucus. Furthermore, it remains unclear how the nanobody acts within the body and how the immune system reacts to nanobody treatment. These challenges are currently being addressed within Collaborative Research Center 1449 “Dynamic Hydrogels at Biointerfaces,” within the framework of which the current results were also generated.
The approach of intracellular nanobody therapy could also be helpful beyond cystic fibrosis for other rare genetic diseases in which protein misfolding plays a role and for which there are currently few effective treatments.
About Cystic Fibrosis
Cystic fibrosis is one of the most common fatal hereditary diseases worldwide. As many as 8,000 children, teens, and adults are living with the disease in Germany today. An imbalance in salt and water levels in the body causes people with cystic fibrosis to produce thick, sticky secretions that harm organs such as the lungs and pancreas. This leads to progressive loss of lung function and shortness of breath, which still significantly lowers life expectancy despite advances in treatment. Some 150 to 200 children are born with this rare disease in Germany each year. A test for cystic fibrosis is part of routine screening for newborns.
Publication: Franz, L., Rubil, T., Balázs, A., Overtus, M., Kemnitz-Hassanin, K., Govaerts, C., Mall, M. A., & Hackenberger, C. P. R.. A cell-permeable nanobody to restore F508del cystic fibrosis transmembrane conductance regulator activity. Nature Chemical Biology 2026. doi: 10.1038/s41589-026-02199-w
Source: Joint Press Release from the Leibniz-Forschungsinstitut für Molekulare Pharmakologie and Charité – Universitätsmedizin Berlin
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect
Innovation / 14.04.2026
Eckert & Ziegler: Metzler Initiates Coverage with a Buy Rating and a Price Target of € 21.00. Upside Potential: 41%
B. Metzler seel. Sohn & Co. AG (Bankhaus Metzler) has started research coverage of Eckert & Ziegler SE (ISIN DE0005659700), a leading supplier of isotope-based components for nuclear medicine and measurement technology, with a buy recommendation and a price target of € 21.00. This corresponds to an upside potential of 40.8% compared to the Xetra closing price of € 14.92 on April 13, 2026.
Bankhaus Metzler thus highlights the strong market position of Eckert & Ziegler SE, which is benefiting from rising demand for diagnostic and therapeutic radioisotopes in nuclear medicine.
About Eckert & Ziegler.
Eckert & Ziegler SE with more than 1,000 employees is a leading specialist for isotope-related components in nuclear medicine and radiation therapy. The company offers a broad range of services and products for the radiopharmaceutical industry, from early development work to contract manufacturing and distribution. Eckert & Ziegler shares (ISIN DE0005659700) are listed in the TecDAX index of Deutsche Börse.
Contributing to saving lives.
Quelle: https://www.ezag.com
Research, Patient care / 13.04.2026
First “protein map” of neurons that initiate pain
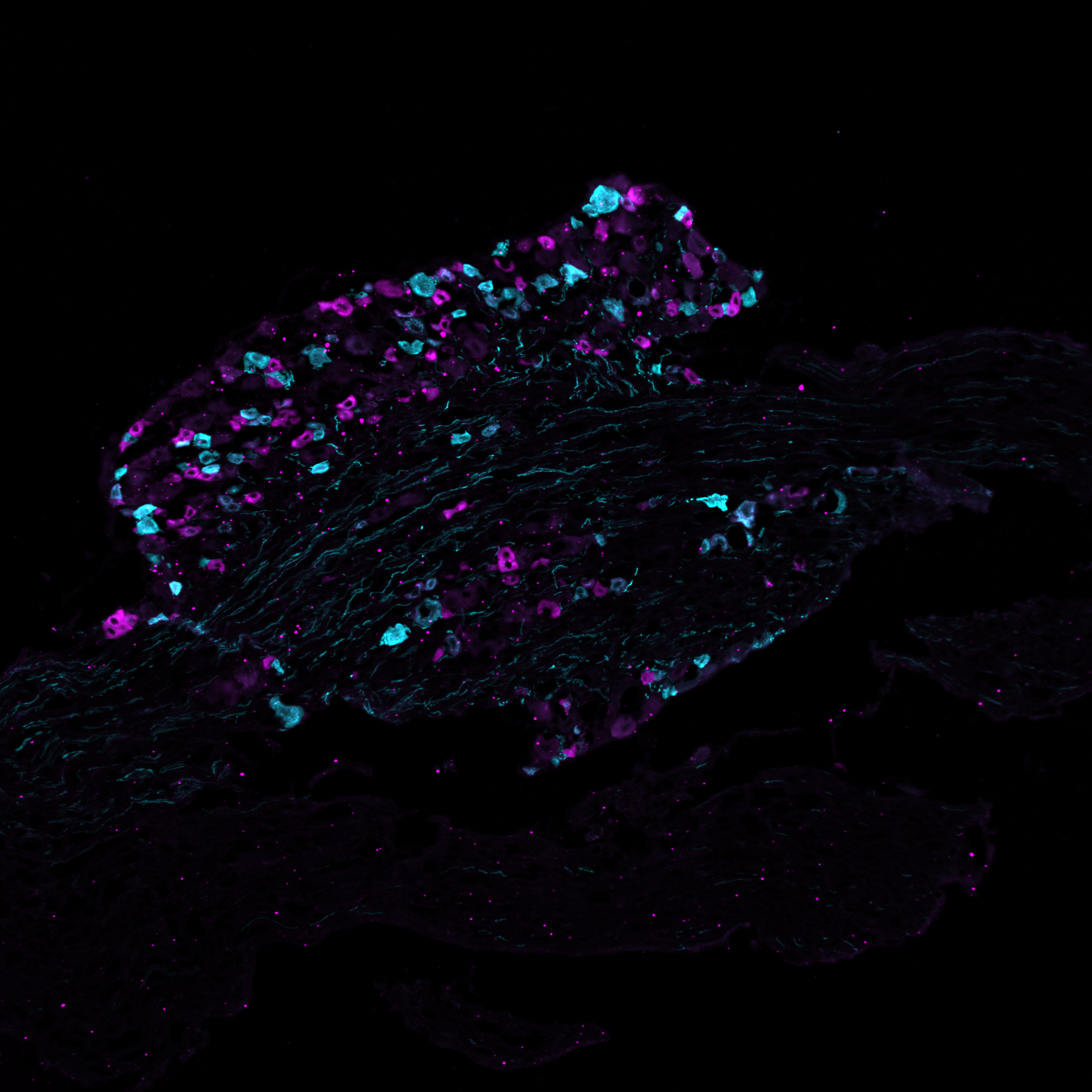
Helmholtz researchers have created the first detailed protein map of specific sensory neurons that trigger pain. Their study, published in “Nature Communications,” will help researchers better understand the molecular mechanisms of chronic inflammatory pain and identify new drug targets.
Joint press release by the Max Delbrück Center and the Helmholtz Centre for Infection Research
One in five people worldwide suffers from chronic inflammatory pain. Meanwhile, about two thirds of those affected find little relief from existing pain medications; new therapeutic approaches are urgently needed. “We first must understand precisely how sensory nerve cells trigger pain at the molecular level — in other words, which proteins are involved,” says Professor Gary Lewin, Group Leader of the Molecular Physiology of Somatosensory Perception lab at the Max Delbrück Center in Berlin.
To unravel these molecular processes, Lewin – who has been studying pain for four decades and recently discovered a previously unknown ion channel involved in pain perception – is working closely with systems biologist Dr. Fabian Coscia, Group Leader of the Spatial Proteomics lab at the same center. Coscia co-developed a method called Deep Visual Proteomics that makes it possible to determine the proteome — the complete set of proteins — of specific cells and to create maps detailing the spatial locations of individual proteins.
The researchers combined this technology with electrophysiological methods from Lewin’s group. This enabled them to first identify specific subtypes of pain neurons based on their function and then analyze their protein profiles. The result is a high-resolution molecular map of these nerve cells, which has been published in “Nature Communications.” The team also demonstrated how the technology can identify potential new drugs targets to treat chronic pain.
Dr. Sampurna Chakrabarti is the study’s first author and a former postdoctoral researcher in the Lewin lab who now heads the Pathways in Infection and Nociception group at the Helmholtz Centre for Infection Research in Braunschweig. Nociception refers to how our nerves respond to stimuli that trigger pain. Nerves in skin and other peripheral tissues – such as muscles and joints – that detect damaging stimuli are called nociceptors; they relay signals to the brain to initiate pain.
Undiscovered signaling pathways
All nociceptors are not alike. “Until now, only the transcriptome – that is, the RNA level information of the different subsets of nociceptors – was known,” says Chakrabarti. “However, the actual functional components of all cells are the proteins formed from these transcripts – and we have now examined them in greater detail for the first time in two subtypes of nociceptors.” Using an electrophysiological method known as the patch-clamp technique, the team first identified and characterized two nociceptor subtypes – peptidergic and non-peptidergic – in the spinal ganglia of mice. Each of these subtypes respond differently to similar stimuli and may initiate pain of different quality and duration.
The researchers used around 50 neurons of each subtype to generate a specific protein map for each of the two cell types. Deep Visual Proteomics combines mass spectrometry with microscopy, artificial intelligence and robotics. Coscia and his team have so far mainly used this methodology for proteome analyses of cancer cells. “We have now shown for the first time that it can also be applied to nerve cells,” he says.
The team measured more than 6,000 proteins in these 50 neurons. A comparison with existing RNA data revealed that the transcriptome and proteome of the cells differ significantly in some cases – an indication that key functional processes only become visible at the protein level. “We provide a unique molecular map of pain-initiating neurons,” says Coscia. “It enables the identification of signaling pathways in these cells that have so far remained hidden.”
In an additional step, Chakrabarti and her colleagues wanted to understand which proteins sensitize nerve cells, contributing to chronic pain. They isolated both types of nociceptors from mouse dorsal root ganglia and exposed them to a molecule called Nerve Growth Factor (NGF), which is known to trigger chronic pain both animals and humans, such as in arthritis. Using Deep Visual Proteomics, the researchers were able to precisely identify the proteins produced after the cells were exposed to NGF.
Reduced sensitivity to pain signals
Lewin and his team had already discovered that NGF plays an important role in chronic inflammatory pain more than 30 years ago. “In dogs and cats, pain can now be alleviated very effectively using antibodies that inhibit NGF,” says Lewin. “In humans, rare side effects have unfortunately prevented their use,” he adds. “But now we may have found an alternative approach: targeting a downstream protein responsible for NGF’s sensitizing effect.”
“We identified several proteins that were present in higher levels in a subset of nociceptors following treatment with NGF. The higher levels of these proteins could be linked to long term pain associated with inflammation,” says Chakrabarti. One of the proteins, an enzyme called B3GNT2, stood out in particular. “When we knocked out the corresponding gene in the cells, the inflammation-induced hyperactivity of nociceptors was reduced. Fewer cells responded to mechanical stimulus,” she says. In other words, the neurons had become less sensitive and would elicit much less pain.
In the future, the researchers plan to validate their findings in mice and humans. “More than 90 percent of all approved drugs now target proteins,” says Coscia. “This highlights how important it is to develop a better understanding of these molecules in order to identify new targets for more effective pain therapies and treatments for other neurological diseases.”
Text: Anke Brodmerkel
Further information
Molecular Physiology of Somatic Sensation
- Chakrabarti lab
- Profile of Gary Lewin
- Profile of Fabian Coscia
- Creating protein maps of tumors
- Understanding the roots of chronic pain
Literature
Sampurna Chakrabarti, Anuar Makhmut, Atena Mohammadi et al. (2026): “Deep visual proteomics uncovers nociceptor diversity and pain targets.” Nature Communications, DOI:10.1038/s41467-026 – 71418‑8
Innovation / 10.04.2026
Eckert & Ziegler Secures Patient Access to Critical Eye Cancer Treatment with MDR Certification for Ru-106 Eye Applicators
Eckert & Ziegler BEBIG GmbH, subsidiary of Eckert & Ziegler SE with focus on brachytherapy solutions for the treatment of eye tumors and prostate cancer, obtained the MDR certificate for its Ruthenium-106 (Ru-106) Eye Applicators from competent authorities. Eckert & Ziegler is the only global provider of these eye applicators. Therefore, this important milestone is a critical safeguard against treatment shortages.
The Medical Device Regulation (MDR) is a European Union directive (EU 2017/745) with the aim of improving the quality of medical devices and increasing patient safety. Obtaining this certification guarantees the long-term availability of Ru-106 Eye Applicators within the EU. These medical devices have been manufactured and internationally marketed by Eckert & Ziegler for more than 30 years. They are actively used in almost 50 countries and contribute several million euros in annual sales to the Eckert & Ziegler Group's earnings.
During an ophthalmic brachytherapy procedure, a small radioactive plaque containing Ru-106 is used to treat uveal melanoma in adults or retinoblastoma in children. The plaque is sutured to the wall of the eye, adjacent to the tumor, and left in place for several days until the required dose of radiation has been delivered. As an alternative to the removal of the affected eye, this treatment offers a chance for patients to conserve vision and quality of life.
"The dedication of our team to achieve MDR certification for a niche product like the Ru-106 Eye Applicators demonstrates our commitment to ensure the long-term availability of this vital form of therapy to treatment centers and patients," explained Katrin Antonenko, Managing Director of Eckert & Ziegler BEBIG GmbH. “The milestone furthermore marks the durable legacy of a product that started to pave the way for the sustainable and successful growth of the Eckert & Ziegler Group more than three decades ago. The experience gained during the successful approval process is expected to expedite additional projects.”
About Eckert & Ziegler
Eckert & Ziegler SE, with more than 1,000 employees, is a leading specialist in isotope-related components for nuclear medicine and radiation therapy. The company offers a broad range of services and products for the radiopharmaceutical industry, from early development work to contract manufacturing and distribution. Eckert & Ziegler shares (ISIN DE0005659700) are listed in the TecDAX index of Deutsche Börse.
Source: Press Release Eckert & Ziegler
Eckert & Ziegler Secures Patient Access to Critical Eye Cancer Treatment with MDR Certification for Ru-106 Eye Applicators
Research / 08.04.2026
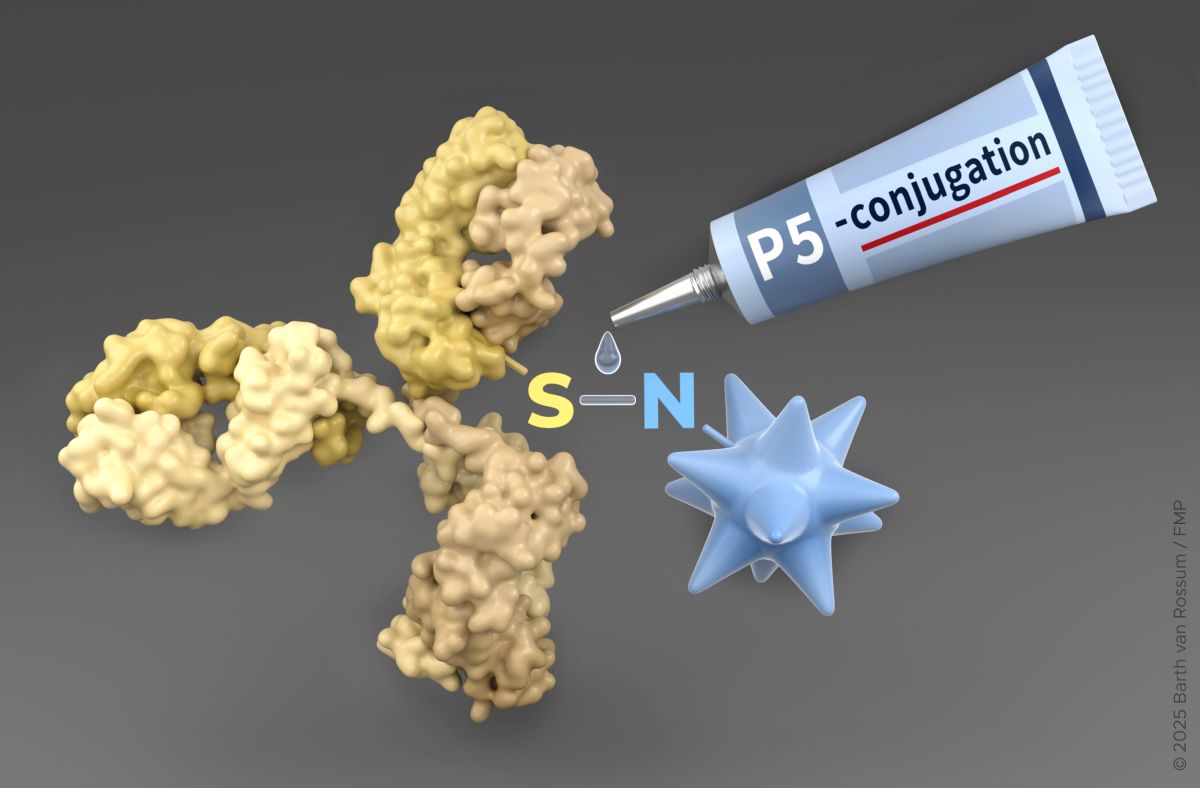
Gilead acquires FMP and LMU Spin-off Tubulis and expands Oncology Pipeline with next-generation ADC
U.S. biopharmaceutical company Gilead has entered into a definitive agreement to acquire Tubulis GmbH. Tubulis was spun off in 2019 from the Leibniz Forschungsinstitut für Molekulare Pharmakologie (FMP) and LMU Munich and develops next-generation antibody-drug conjugates (ADCs) designed to deliver various active ingredients more selectively to tumors and maximize patient benefit. The transaction expands Gilead’s oncology pipeline with several innovative programs and platform technologies, some of which originated from academic research at the Leibniz Forschungsinstitut für Molekulare Pharmakologie (FMP) in Berlin.
The acquisition includes Tubulis’ lead product, TUB-040, a NaPi2b-targeted topoisomerase I inhibitor ADC currently in Phase 1b/2 clinical development for the treatment of platinum-resistant ovarian cancer and non-small cell lung cancer (NSCLC). Gilead will also acquire TUB-030, a 5T4-targeted ADC with promising early clinical data across various solid tumor types as well as Tubulis’ next-generation ADC platform and a promising early pipeline.
Dr. Dominik Schumacher, CEO and co-founder of Tubulis, also emphasizes the importance of combining scientific excellence with industrial development strength: „From the outset, we believed our conjugation technology platforms could have broad impact across the ADC field and the initial data from TUB-040 have reinforced that conviction,” said Dr. Dominik Schumacher, Chief Executive Officer and Co-founder of Tubulis. „Joining Gilead allows us to build on this foundation within an organization that brings deep scientific expertise, global development capabilities, and the scale needed to translate innovation into medicines for patients worldwide. Through our existing collaboration, Gilead has already seen the potential of our technologies and together, we are well positioned to accelerate the development of our ADC pipeline. I’m deeply grateful to the Tubulis team, our Board of Directors, investors, and partners for their commitment and helping make this milestone possible.”
P5 Conjugation Technology from FMP
A key role in Tubulis’ product development is played by the innovative P5 conjugation technology, a linker chemistry that enables the precise coupling of antibodies with highly potent active ingredients. It was developed through basic research by Prof. Dr. Christian Hackenberger, one of Tubulis’ co-founders, and his team at the Leibniz Forschungsinstitut für Molekulare Pharmakologie (FMP). Tubulis has further developed P5 technology for clinical research and application in collaboration with the research group led by Prof. Dr. Heinrich Leonhardt (Ludwig Maximilian University of Munich), a co-founder of Tubulis. “This successful technological development demonstrates the potential of interdisciplinary collaboration across departmental and institutional boundaries”, says Prof. Dr. Heinrich Leonhardt. This technology forms a central platform for stable and controlled drug delivery in the company’s ADC programs. „This acquisition not only underscores the success of our conjugation technology and the performance of the Tubulis team in improving cancer therapy with new ADCs. It also demonstrates the importance and impact of basic research at universities and research institutes for translational innovation", says Prof. Dr. Christian Hackenberger.
Upon completion of the transaction, Tubulis will operate as an independent ADC research organization within Gilead, with the Munich site serving as a hub for ADC innovations. The company will build on existing integrated capabilities in research, production, and clinical development to advance next-generation ADCs.
Gilead will acquire all of the outstanding equity of Tubulis for 3.15 billion US Dollars in upfront cash consideration on a cash-free, debt-free basis, plus up to 1.85 billion US Dollars in contingent milestone payments. The acquisition is expected to close in the second quarter of 2026. Closing of the transaction is subject to expiration or termination of certain regulatory filings and other customary closing conditions.
About Tubulis
Tubulis develops tailored antibody-drug conjugates (ADCs) with improved biophysical properties. In preclinical models, the ADCs have already demonstrated targeted and sustained accumulation in the tumor as well as long-lasting anti-tumor effects. The two most advanced programs in the growing pipeline are TUB-040 (targeting NaPi2b) and TUB-030 (targeting 5T4). Both programs are currently being investigated in clinical trials for cancers with high unmet medical needs. For more information, visit: www.tubulis.com
Source: Leibniz-Forschungsinstitut für Molekulare Pharmakologie
Gilead acquires FMP and LMU Spin-off Tubulis and expands Oncology Pipeline with next-generation ADC
Research / 27.03.2026
Friedrich Stolz Award for the Tubulis Team

The Medicinal Chemistry Division of the Gesellschaft Deutscher Chemiker (GDCh) and the Pharmaceutical/Medicinal Chemistry Division of the Deutsche Pharmazeutische Gesellschaft (DPhG) have awarded the Friedrich Stolz Award 2026 to the team from Tubulis, a spin-off of the Leibniz-Forschungsinstitut für Molekulare Pharmakologie and LMU Munich. Honored are Prof. Dr. Christian Hackenberger from the Leibniz-FMP as co-founder and advisor and Dr. Jonas Helma-Smets, co-founder and CSO, Dr. Marc-André Kasper, VP Chemistry and Early Discovery and Dr. Annette Vogl, VP Biology and Translational Research, from Tubulis.
The Friedrich Stolz Award recognizes exceptional dedication, scientific discoveries, or innovative technologies with proven relevance that contribute to substantial therapeutic innovation or sustainable development and go beyond early research stages.
The Tubulis team was awarded for their P5 ethynylphosphonamidate conjugation chemistry, enabling a novel type of antibody-drug conjugates (ADCs).The conjugation chemistry, also termed P5-labeling, was developed at FMP in the research unit of Christian Hackenberger. This new ADC class is characterized by exceptional plasma stability, favorable pharmacokinetic properties, and pronounced anti-tumor efficacy. Building on this, NaPi2b-targeting exatecan ADCs with particularly stable linkers, reduced off-target toxicity, and optimized molecular architecture were generated. The culmination of this work is the development candidate TUB-040, which in a clinical Phase 1 study in patients with platinum-resistant ovarian cancer demonstrates promising tumor remissions, a wide therapeutic window, and good tolerability. This work shows how innovative chemistry can be directly translated into novel, potentially more effective treatment options for hard-to-treat cancers.
The prize is awarded in memory of industrial pharmacist and drug researcher Friedrich Stolz, whose visionary innovative power serves as a model for application-oriented, therapeutically relevant research.
Photo: Awarded the Friedrich Stolz Prize: Prof. Dr. Stefan Laufer (GDCh), Prof. Dr. Christian Hackenberger, Dr. Annette Vogl, Dr. Jonas Helma-Smets and Dr. Franz von Nussbaum (from left to right). © GDCh
Source: FMP
Friedrich Stolz Award for the Tubulis Team
Innovation / 26.03.2026
Eckert & Ziegler: Strong FY 2025 with Positive Outlook
Eckert & Ziegler SE (ISIN DE0005659700, TecDAX) set a new record in fiscal year 2025 with sales of €312.0 million. Compared to the previous year, sales rose by approximately €16 million (+5%). EBIT before special items from continuing operations (adjusted EBIT) increased by just under €12 million year-over-year to €77.7 million (+18%). Net income rose by approximately €15 million (+46%) to €48.8 million; this corresponds to earnings per share of €0.78 (previous year: €0.53, adjusted for stock split).
In the Medical segment, sales increased by €22.6 million, or 15%, to €171.3 million. The business with pharmaceutical radioisotopes remains the most important source of revenue.
The Isotope Products segment generated sales of €150.1 million, a decrease of €7.9 million (-5%) compared with the previous year. This was mainly due to a temporary shift toward lower-margin products compared with the same period last year. In addition, the cyberattack in 2025 led to project-related delays.
For the 2026 fiscal year, the Executive Board expects an adjusted EBIT of approximately €80 million. The corresponding sales forecast amounts to approximately €320 million. This forecast is based on a weighted average exchange rate of $1.20 per euro. Adjusted for currency effects and the licensing business, this corresponds to a growth of 9% in sales and 21% in earnings (adjusted EBIT).
The Executive Board and Supervisory Board will propose to the Annual General Meeting a dividend of €0.22 (previous year: €0.17, adjusted for stock split) per share entitled to dividends.
The 2025 financial statements can be found here: https://www.ezag.com/fy2025en/
Overview
FY 2025:
- Sales: €312.0 million (previous year: €295.8 million)
- adjusted EBIT: €77.7 million (previous year: €65.9 million)
- Net income: €48.8 million (previous year: €33.3 million)
Forecast 2026:
- Sales of approx. €320 million
- adjusted EBIT of approx. €80 million
About Eckert & Ziegler.
Eckert & Ziegler SE with more than 1.000 employees is a leading specialist for isotope-related components in nuclear medicine and radiation therapy. The company offers a broad range of services and products for the radiopharmaceutical industry, from early development work to contract manufacturing and distribution. Eckert & Ziegler shares (ISIN DE0005659700) are listed in the TecDAX index of Deutsche Börse.
Contributing to saving lives.
Research, Innovation, Patient care / 23.03.2026
Launch of the Einstein Center for Early Disease Interception

At the Einstein Center for Early Disease Interception, researchers from twelve Berlin institutions will be pooling their expertise to explore new approaches to prevention, long before symptoms appear, and bring them quickly into practice. The center opens today with a ceremony
Joint press release by the Max Delbrück Center, Charité – Universitätsmedizin Berlin, Berlin Institute of Health at Charité, and Technische Universität Berlin
Serious diseases often develop unnoticed over many years. By the time symptoms appear, organ damage is often too extensive to be reversed completely. In many cases, there are no effective treatments to stop diseases from progressing.
Researchers from twelve leading Berlin institutions are heading the charge to change the way we think about prevention. Using the latest technologies – including advances made by researchers from Berlin – they aim to unravel what happens inside cells at the very earliest stages of disease. Such knowledge will enable them to develop early interventions, when only individual cells are affected and diseases are still controllable.
The Einstein Center for Early Disease Interception (EC-EDI) is bringing together the necessary expertise to study such early disease mechanisms. The center is being officially inaugurated today at the Max Delbrück Center’s Berlin Institute for Medical Systems Biology in Mitte after a two-year preparatory phase. Invited guests include politicians, scientists and the public. The Einstein Foundation Berlin is funding the center with €6 million and is supporting the recruitment of young international talent with additional funds from the state of Berlin.
A dynamic network for key technologies
Over the next six years, researchers will further develop, integrate, and apply key technologies to the effort. These include single-cell multiomic and spatial biology technologies, patient-specific organoids, 3D bioprinting, and AI-based modeling of mechanisms and disease trajectories. A platform for cross-institutional collaboration will also be established to help translate research findings into clinical practice more quickly in partnership with industry and investors – while generating societal and economic value for the state of Berlin. Initial focus areas include respiratory and neurological diseases, such as inflammatory lung diseases, tuberculosis, Alzheimer’s disease, and multiple sclerosis.
Participants in EC-EDI include Charité – Universitätsmedizin Berlin, the Berlin Institute of Health at Charité, the Max Delbrück Center, Technische Universität Berlin, Freie Universität Berlin, Humboldt-Universität zu Berlin, and additional non-university institutions such as the Museum für Naturkunde and several Max Planck Institutes.
Statements from Einstein Center spokespersons
Professor Nikolaus Rajewsky (Head Spokesperson; Director of the Berlin Institute for Medical Systems Biology at the Max Delbrück Center and Professor at Charité):
“We are advancing molecular prevention and intervention – and aim to bring our research to patients as quickly as possible. Major breakthroughs do not arise solely in the clinic, in labs, or on a computer. The Einstein Center creates a shared, open platform with clear rules and short pathways. Clinicians, basic researchers, and data scientists will collaborate across institutions, as well as with companies and investors. This will enable us to work faster and will hopefully help further establish Berlin as a leading international hub in this field.”
Professor Leif Erik Sander (Project Head; Director of the Department of Infectious Diseases and Critical Care Medicine at Charité and Research Group Leader at BIH):
“Conventional methods often fail to detect the early signs of emerging diseases. Moreover, we don’t always understand what is actually going wrong at the molecular level or how we could intervene to stop these diseases. This is where early disease interception comes in: Using high-resolution technologies, we aim to understand what sets things on the wrong track and create opportunities to correct that course very early on. The train, so to speak, needs to be put back on the track to health. We want to use our findings to develop innovative solutions, such as new diagnostic tests and medications. This is how research leads to medical progress and value creation – and that strengthens Berlin as a hub for innovation and business.”
Dr. Janine Altmüller (Head of the Core Unit Genomics at the Berlin Institute of Health at Charité, BIH):
“New methods are key to the Einstein Center’s vision: Never before has it been possible to examine characteristic features such as nucleic acids or proteins of individual cells in their natural tissue context with such precision and high resolution. Even the smallest changes that mark the onset of a disease can now be digitalized and, with the help of AI for example, better deciphered and understood. This knowledge is crucial for developing targeted interventions. At the Einstein Center, experts from various disciplines will be working together toward this goal, thereby accelerating the translation of scientific findings into clinical applications.”
Professor Jens Kurreck (Executive Director of the Institute of Biotechnology, Technische Universität Berlin):
“The Einstein Center for Early Disease Interception provides a unique platform for close collaboration among leading Berlin-based research institutions and enables a better understanding of the early mechanisms of disease development using various human models. It is particularly important to offer young scientists access to state-of-the-art technologies within this consortium and to create optimal conditions for them to build their scientific careers.”
Further information
Research, Innovation, Patient care / 19.03.2026
Strengthen international cooperation in clinical trials

As part of the Australian Clinical Trials Initiative, ACTI, Australian companies and representatives of the Australien Government’s Trade and Invest Commission, Austrade, visited the Berlin-Buch Science and Biotech Campus today. The initiative promotes Australia as an attractive location for clinical trials.
Dr. Christina Quensel, CEO der Campus Berlin-Buch GmbH, welcomed the guests and, during a tour, provided information about the campus’s ecosystem of basic biomedical and clinical research, life science startups, and biotechnology companies. Biotech and medtech companies operate here in one of Europe's leading biotech parks.
The delegation gained insight into two scientific core facilities the Proteomic Lab of the Berlin Institute of Health in the Charité und the Cryo Electron Microscopy, a collaborative scientific infrastructure involving Charité – Universitätsmedizin Berlin, the Max Delbrück Center, and the Leibniz Research Institute for Molecular Pharmacology.
At the BerlinBioCube startup center, the guests visited the startups CheckImmune GmbH, a CRO, and T-knife GmbH, a pharmaceutical company that develops highly innovative cancer therapies.
During a networking lunch, the delegation engaged in intensive discussions with executives and stakeholders of the BiotechPark Berlin-Buch.
Many thanks to our guests for the exciting exchange!
Minesh Lalla, Senior Business Development Manager at Peter MacCallum Cancer Centre (CTS)
Jason Erasmus, Business Development Manager at Beyond Drug Development (CRO)
Tim Vale, Business Development Manager at Resolian Bioanalytics (Lab)
Karl Boehle, Senior Business Development Manager, Austrade
Pallavi Mishra, South Australia Trade & Investment Manager, Austrade
George Zafiris MBA, Prime Financial Group (R&D Tax Advisors)
Bellberry (Ethics)
The delegation then proceeded to Berlin Partner for Technology and Business, which, in cooperation with Austrade, had hosted an exclusive seminar on opportunities for German-Australian collaboration in clinical trials. The event highlighted how the Australian clinical trial ecosystem is structured and the key strengths and efficiency advantages it offers. Executives were invited The event was aimed at pharmaceutical and biotech R&D leaders, clinical operations and trial strategy decision-makers, CROs and clinical service providers, as well as leaders from academic, hospital, and research institutions, and stakeholders in policy, innovation, and the ecosystem.
Research / 17.03.2026
Fat tissue fuels gut inflammation
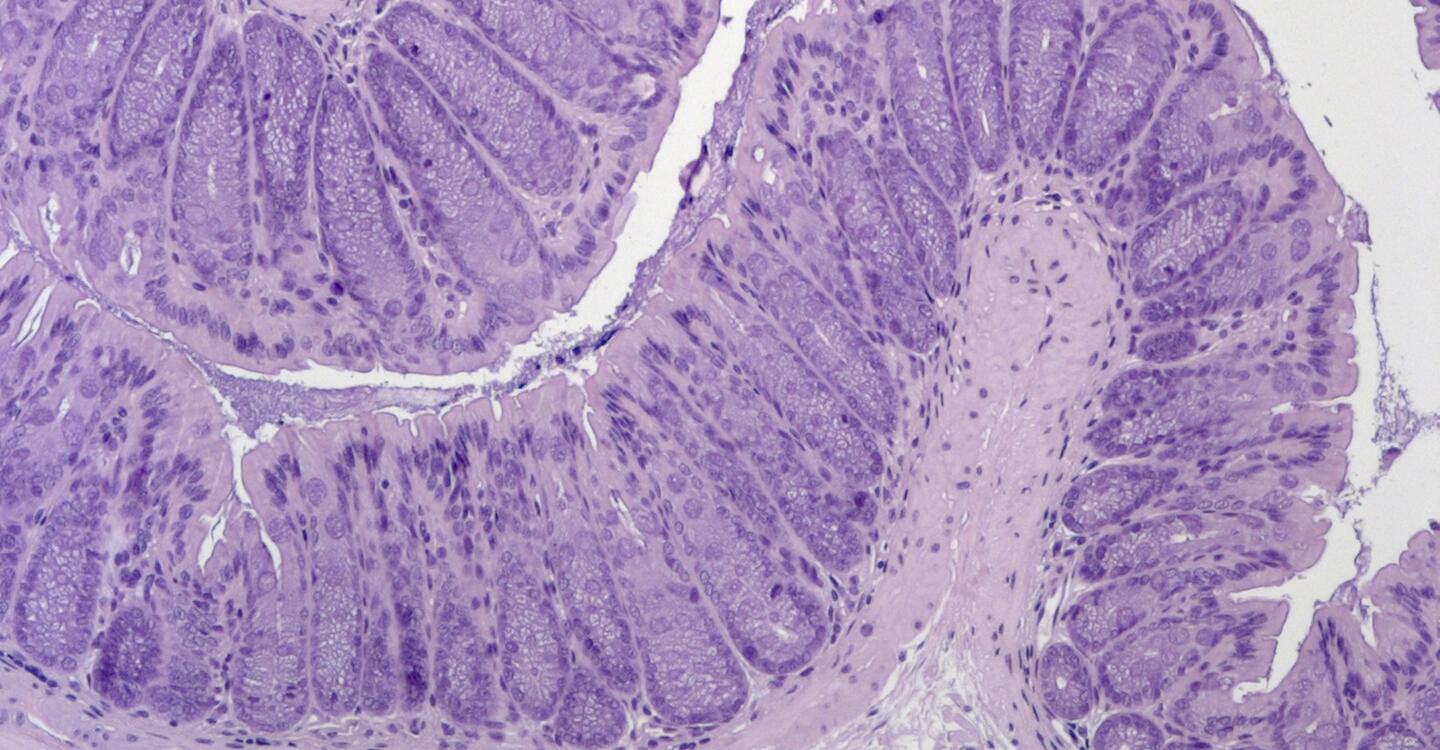
Researchers at the Max Delbrück Center and Charité – Universitätsmedizin Berlin show that adipose tissue amplifies gut inflammation in Crohn’s disease. The study, published in the “Journal of Clinical Investigation,” also reveals how genetic changes in immune cells may contribute to the disease.
Crohn’s disease is a chronic inflammatory disorder of the intestinal tract affecting millions of people worldwide. One prominent feature of the disease is “creeping fat” — adipose tissue that wraps around inflamed sections of the intestine. Scientists have long suspected that this fat tissue may influence disease progression, but the underlying mechanisms have remained unclear
Now, researchers from Charité – Universitätsmedizin Berlin, the Berlin Institute for Medical Systems Biology at the Max Delbrück Center (MDC-BIMSB) and their collaborators show that fat tissue releases signals that ultimately damage the intestinal barrier in Crohn’s disease. They also show that acquired genetic mutations in immune cells may fuel the disease.
“There is a longstanding link between obesity and Crohn’s disease,” says Dr. Ashley Sanders, Group Leader of the Genome Instability and Somatic Mosaicism Lab at the Max Delbrück Center and a senior author of the paper. “While it’s fairly well established that adipose tissue secretes molecular signals that promote inflammation, here we detail the link between fat-derived signals and intestinal inflammation.”
Mice with no fat tissue
The researchers first conducted experiments in a mouse model of generalized lipodystrophy – an extremely rare disease in which affected people lack fat tissue among other symptoms.
When they exposed the mice to a chemical substance known to cause intestinal inflammation, they found the lipodystrophy mice were more resistant than normal mice. Their intestinal barrier remained stable and they had low levels of pro-inflammatory immune cells, particularly Th1 and Th17 T cells – specialized subsets ofimmune cells that coordinate immune responses.
When the team transplanted adipose tissue back into the lipodystrophy mice, they became more susceptible to the chemically induced gut inflammation. Remarkably, they observed this effect only when the transplanted fat was capable of producing leptin, suggesting that leptin plays a central role in regulating the immune system in mice.
“These findings were expected because previous research had already established that fat tissue is an active immune organ,” says Dr. Britta Siegmund, a senior author of the paper at Charité — Universitätsmedizin Berlin. “But they nevertheless highlight that signals derived from adipose tissue can substantially amplify intestinal inflammatory responses.”
Puzzling clinical findings
However, the results appeared to conflict with findings from a clinical case at Charité: A patient with acquired generalized lipodystrophy who had developed Crohn’s disease, despite having no fat tissue at all. “This was curious and we wanted to understand why this patient had developed the disease,” Siegmund adds.
Using single-cell sequencing, the Sanders lab examined immune cells extracted from the patient’s blood and intestinal tissue. The analyses showed an unusually large group of cells that carried a mutation in a gene called NRAS, which helps regulate cell growth and survival. This suggested that these cells had multiplied from a single original cell in a process called clonal expansion – such mutated cells arise spontaneously and are not inherited. Cells with mutated NRAS survive longer and divide more than they should, potentially driving chronic inflammation.
“This mutation likely helped the inflammatory T cells survive and expand over time,” explains Sanders. “That persistent expansion may have fueled the systemic and intestinal inflammation that was observed. What’s more, our results show how powerful single-cell genomic technologies can be for uncovering hidden genetic changes in individual immune cells,” she adds.
Toward more personalized treatment
Among other pro-inflammatory signals, adipose tissue also secretes mediators called adipokines — including leptin, which is best known for regulating appetite and metabolism. In the study, the lipodystrophy patient was given a synthetic form of leptin to treat type-2 diabetes. While the hormone improved the patient’s metabolic health, it also increased pro-inflammatory signaling and raised levels of Th17 immune cells.
“The finding highlights how adipokines such as leptin and genetic changes in immune cells interact to influence chronic inflammatory diseases,” says Dr. Carl Weidinger, another senior author of the study at Charité. This dual role of metabolic and genetic factors could help explain why Crohn’s disease behaves differently in different people, he adds. “Understanding these interactions may help us develop more precise, personalized approaches to treatment.”
Text: Gunjan Sinha
Further information
Genome instability and somatic mosaicism
Literature
Marilena Letizia, Toka Omar, Patrick Weidner et al. (2026): “Characterization of intestinal immune responses in generalized human and murine lipodystrophy.” Journal of Clinical Investigation, DOI: 10.1172/JCI192322.
Image
Colon tissue from a mouse model of acquired generalized lipodystrophy. Colon tissue remains intact after exposure to a chemical that causes gut inflammation. © Anja Kühl, Charité
Research, Patient care / 13.03.2026
How cancer immunotherapies affect the heart
Checkpoint inhibitors help fight cancer but increase the risk of heart disease. An interdisciplinary team, including researchers from the Max Delbrück Center, is investigating how the therapy can be made safer. The Hector Foundation is supporting the project with €1.3 million.
Checkpoint inhibitors have become standard cancer therapy in recent years. These antibody drugs stimulate the immune system to attack malignant cells more aggressively. They were first widely used to treat patients with melanoma, but are now also indicated for patients with lung, kidney, or bladder cancer because the immunotherapy treatment is more effective than other options.
However, the drugs are not good for the heart. “We estimate that cancer patients receiving immunotherapy with checkpoint inhibitors have about a threefold increased risk of heart disease such as coronary artery disease,” says cardio-oncologist Dr. Lorenz Lehmann from Heidelberg University Hospital. Together with an interdisciplinary team of researchers, Lehmann is studying the biological mechanisms that cause these side effects in an effort to reduce the therapy’s impact on the heart.
The consortium, called ATHERIC-CARE (Atherosclerosis Risk in Cancer Patients after Immune Checkpoint-Inhibitor Therapy – Assessment and Exploration), includes Dr. Isabel Poschke from the German Cancer Research Center in Heidelberg, Dr. Henrike Maatz from the Max Delbrück Center in Berlin-Buch, Professor Sylvia Saalfeld from Kiel University, and Professor Oliver Müller from the University Hospital Schleswig-Holstein in Kiel. Beginning February 1, 2026, the Hector Foundation will fund the multicenter team’s research with €1.3 million over three years.
Inflamed coronary arteries
Coronary artery disease is a form of atherosclerosis affecting the coronary arteries. Lipids and other substances from the bloodstream accumulate on the inner walls of the vessels and trigger chronic inflammation. Over time, the blood vessels become increasingly narrowed, which can lead to heart failure or a heart attack. ATHERIC-CARE is primarily investigating how checkpoint inhibitors activate or accelerate this inflammatory mechanism.
The scientists are using DNA analyses to investigate which immune cells drive atherosclerosis, for example. In animals, they are studying early inflammatory mechanisms and analyzing computed tomography images of affected individuals with assistance from machine learning tools. Such analyses can not only detect metastases, but also reveal changes in the coronary arteries.
“Our task will be to determine which genes become active in the cells of the coronary arteries and in the surrounding tissue – in other words, what exactly is happening at the molecular level in the different cell types,” says Dr. Henrike Maatz, a scientist in the Genetics and Genomics of Cardiovascular Diseases lab headed by Professor Norbert Hübner at the Max Delbrück Center. To find out, she will primarily be using two methods: single-nucleus RNA sequencing (snRNA-seq) and spatial transcriptomics.
Research for better quality of life
“For example, we want to characterize more precisely the interaction between immune cells and endothelial cells, which line the inside of blood vessels,” explains Maatz. “Our goal is to understand how immunotherapy promotes T cell-mediated inflammation in blood vessels, which can lead to unstable vascular deposits and atherosclerosis.” Using spatial transcriptomics, she also aims to determine how immune cells are distributed within plaques. The Hector Foundation is supporting the research with about €286,000.
“By bringing together the results generated in the consortium’s different research areas, we aim to identify biomarkers and strategies that make immunotherapy safer – and thereby improve patients’ quality of life,” says project leader Lehmann.
Maatz is looking forward to the upcoming collaboration: “Together, we will of course achieve much more than a single research group could,” she says. “And we have already worked very successfully with Lorenz Lehmann on other projects.”
Further information
Research / 07.03.2026
Equality in research drives innovation

Marking International Women's Day, Federal Research Minister Dorothee Bär met with female researchers at the Max Delbrück Center
Statement from Federal Research Minister Bär:
“Equality is essential for Germany's capacity for innovation and long-term competitiveness. It is a decisive driver of progress. If we want to address the major challenges and megatrends of our time, be it digitalization, space research, medicine of the future, or key technologies, we cannot afford to leave half of the brightest minds out of driving Germany’s future forward. This is why the Federal Ministry of Education and Research, together with the states, is supporting equality at universities with a 2030 female professors program and the visibility of female researchers with the “Focus on Innovative Women” funding guidelines. In order to further strengthen social awareness of gender issues in research, we are currently designing a federal scientific award for gender research, which is to be announced later this year.”
Statement from Professor Maike Sander, Scientific Director and Chair of the Executive Board of the Max Delbrück Center:
“The discoveries made at the Max Delbrück Center lay the foundation for the medicine of the future. To get there, we need talented individuals who are enthusiastic about innovation and eager to contribute their ideas – regardless of gender or background. A total of 60 percent of our researchers are women. We have also been successful in recruiting early-career female group leaders. However, we still have some catching up to do when it comes to permanent professorships. This is why we specifically promote female scientists, for example through mentoring programs. We also approach exceptional candidates worldwide. More than half of our group leaders have been recruited internationally. We want to create an inspiring environment for all of them. We need a scientific culture in which careers are defined not by traditional role expectations, but by curiosity, talent, and courage. There is still much to be done.”
Photo: From left to right: Maike Sander, Katja Simon, Dorothee Bär and Ashley Sanders in a lab.
Maike Sander is Scientific Director of the Max Delbrück Center. Katja Simon is Head of Scientific Talent Management, Dorothee Bär is Federal Minister of Research, Technology and Space, Ashley Sanders is scientific group leader.
© Felix Petermann, Max Delbrück Center
Source: Max Delbrück Center
Equality in research drives innovation
economic development, Innovation / 03.03.2026
T-knife Therapeutics Announces Authorization of Clinical Trial Application for TK-6302, A Multi-Armored, CRISPR based T cell Therapy for Solid Tumors
Phase 1 ATLAS trial to evaluate TK-6302 in patients with advanced PRAME-positive solid tumors authorized for initiation
SAN FRANCISCO and BERLIN, March 03, 2026 - T-knife Therapeutics, Inc., a biopharmaceutical company developing next-generation T cell therapies to fight cancer, today announced the authorization of its Clinical Trial Application (CTA) to begin the Phase 1 ATLAS trial of TK-6302 in Europe. TK-6302 is a multi-armored PRAME-targeted T cell therapy specifically engineered to overcome the challenges associated with treating solid tumor cancers. The ATLAS trial, which is poised to begin this year, is an adaptive, first-in-human, open-label, Phase 1 trial of TK-6302 in patients with advanced PRAME-positive solid tumors.
“We are proud to achieve this important milestone, which enables the initiation of the Phase 1 ATLAS clinical trial and our transition to becoming a clinical-stage company,” stated Thomas M. Soloway, President and Chief Executive Officer of T-knife. “TK-6302 is a highly differentiated therapy, engineered for greater potency against PRAME, a clinically validated target with attractive commercial potential. PRAME is highly prevalent across multiple high unmet-need solid tumor indications, including squamous non-small cell lung, ovarian, endometrial, skin, and triple-negative breast cancers. Today’s announcement reflects the dedication, expertise, and urgency our team brings to transforming innovative science into life-changing therapies.”
Peggy Sotiropoulou, Ph.D., Chief Scientific Officer of T-knife, added, “The CTA was supported by a comprehensive and compelling preclinical data package demonstrating TK-6302’s best-in-class anti-tumor efficacy. By bolstering T-cell fitness and persistence while overcoming challenging tumor mediated immune barriers, TK-6302 has the potential to deliver meaningful clinical benefit, including deep, durable responses across a range of solid tumor cancers. Bringing a first-of-its-kind multi-armored, CRISPR gene-edited T cell therapy into the clinic underscores our commitment to pushing scientific boundaries in the service of delivering transformative therapies to patients.”
About TK-6302
TK-6302 is a PRAME-targeted T cell therapy that has been “supercharged” by the inclusion of multiple armoring innovations: a high affinity PRAME targeting receptor to enhance cytotoxicity; a costimulatory CD8 coreceptor to engage CD4 T cells and enhance T cell fitness and persistence; and a FAS-based checkpoint converter designed to boost engraftment and promote T cell survival in the hostile tumor micro-environment. Preclinical data with TK-6302 demonstrated sustained serial killing and cytokine secretion in a model mirroring the inhibitory ligand expression in PRAME-expressing tumors. In a complex 3-dimensional (3D) spheroid tumor model, TK-6302 eliminated multiple rounds of tumors and demonstrated superior anti-tumor activity compared to controls. TK-6302 is manufactured with a non-viral gene editing process for improved T cell receptor expression, and it has been successfully manufactured at-scale using the clinical process.
About T-knife Therapeutics
T-knife is a biopharmaceutical company dedicated to developing T cell therapies to deliver broad, deep and durable responses to solid tumor cancer patients. The company’s unique approach leverages its proprietary platforms and synthetic biology capabilities to design the next-generation of supercharged T cells with best-in-class potential.
T-knife was founded by leading T cell and immunology experts utilizing technology developed at the Max Delbrück Center for Molecular Medicine together with Charité – Universitätsmedizin Berlin, is led by an experienced management team, and is supported by a leading group of international investors, including Andera Partners, EQT Life Sciences, RA Capital Management and Versant Ventures. For additional information, please visit the company’s website at www.t-knife.com.
www.t-knife.comResearch / 25.02.2026
Gut microbes affect the heart via the brain
Researchers at the Max Delbrück Center have uncovered how a bacterial metabolite produced in the gut regulates heart function through specific brain cells. The findings, published in “Circulation Research,” reveal a new gut-brain-heart axis and point to potential therapies for hypertension and heart failure.
Hypertension and heart failure affect millions worldwide. Yet in many patients, doctors cannot fully explain why the heart becomes stiff and struggles to relax – a condition known as diastolic dysfunction.
Researchers in the lab of Dr. Suphansa Sawamiphak, Group Leader of Cardiovascular-Hematopoietic Interaction at the Max Delbrück Center, have identified a direct communication line between gut bacteria, the brain, and the heart. Using zebrafish as a model, the team discovered that certain gut microbes produce a small molecule called indole-3 acetic acid (IAA) from the dietary amino acid tryptophan. IAA acts on neurons in the brain, which in turn, control the heart. The study was published in “Circulation Research.”
“We were surprised that a single bacterial metabolite could influence the central nervous system, the heart, and major hormonal systems at the same time,” says Bhakti Zakarauskas-Seth, lead author of the paper. “It shows that the brain can act as a central hub in gut-heart communication.”
Tracking a signal from gut to brain
To understand how gut bacteria might influence the heart, the researchers focused on a distinct group of neurons in the hypothalamus known as hypocretin (Hcrt) in zebrafish larvae. These cells produce Hcrt neuropeptides, also known as orexins, which regulate many involuntary functions in the body – such as sleep and hunger – and also heart activity. When IAA levels dropped, Hcrt neurons became overactive. This increased sympathetic nerve signals to the heart, causing the heart muscle to stiffen, impairing its ability to relax properly.
When the researchers supplemented the larvae with IAA, neuronal activity normalized, heart function and blood pressure improved, and even related hormones such as renin and angiotensinogen returned to healthier levels.
They then examined data from a cohort of patients – humans also have Hcrt neurons – and found that IAA levels were reduced in patients with hypertension. Notably, they observed a sex-specific effect, with hypertensive women showing significantly lower levels of IAA in their serum samples than men.
Implications for patients and prevention
Diastolic dysfunction very common – up to half of all people over age 70 have some level of impairment. It is also is the underlying functional mechanism of heart failure with preserved ejection fraction (HFpEF), which accounts for over 50% of all heart failure cases.
For these patients, the findings open several potential avenues for better care, says Zakarauskas-Seth. “IAA levels could serve as a biomarker to identify patients at high risk of hypertension or heart failure. Therapeutically, boosting IAA production – for example through diet, probiotics, or supplementation – could become a new strategy to prevent or treat cardiovascular disease.”
That a single bacterial metabolite can influence the central nervous system, the heart, and major hormonal system also underscores a broader message, she adds. “The body does not operate in isolated compartments. Gut health, microbial balance, and diet directly shape how well the heart functions.” The researchers will need to validate their findings in other animal models and clinical studies will be needed to determine whether restoring IAA can benefit patients.
Text: Gunjan Sinha
Photo: Rückenansicht einer Zebrafischlarve: Aktive Nervenzellen sind grün, Hypocretine herstellende Neurone und ihre Ausläufer rot dargestellt. © Bhakti Zakarauskas-Seth, AG Sawamiphak, Max Delbrück Center
Source: Press Release Max Delbrück Center
Gut microbes affect the heart via the brain
Research / 24.02.2026
Similar kinases play distinct roles in the brain
Researchers at the Max Delbrück Center show that two closely related enzymes, MNK1 and MNK2, act on the brain to regulate different behaviors. Their findings, published in “Molecular Psychiatry,” could help guide development of more precise therapies for neurological disorders.
Structurally, they look similar: MNK1 and MNK2 belong to the same enzyme family and are best known for regulating how cells make proteins. Their starring role in such a crucial cellular function has cast them into the spotlight as potential drug targets to treat nervous system disorders and chronic pain. But would it matter whether a drug targets only one of them?
In a study published in “Molecular Psychiatry,” researchers led by Rosalba Olga Proce, a doctoral student in the Molecular and Cellular Basis of Behavior lab led by Dr. Hanna Hörnberg at the Max Delbrück Center, set out to determine whether the two enzymes – also called kinases – perform distinct functions in the brain. The team found clear differences. Mice lacking MNK1 showed less interest in newly introduced objects than controls and impaired memory of objects. By contrast, mice without MNK2 appeared normal in object recognition tests but showed enhanced interest in social contacts.
“The behavioral differences we observed suggest that each kinase has a specialized function,” says Proce. “It might be preferable to target each kinase individually when designing drugs.”
Mouse behavior and molecular data
To uncover how these behavioral changes arose, the researchers used genetically engineered mice in which either the MNK1 or MNK2 gene was knocked out. They then combined behavioral testing with large-scale molecular analyses of brain tissue.
The team focused in particular on the cerebral cortex and on synapses – contact points where neurons communicate. Using proteomics, transcriptomics, and phosphoproteomics, they compared protein levels, gene expression, and protein phosphorylation patterns between the two types of mice.
Mice lacking MNK1 had an abundance of ribosomal proteins, which play a central role in transcribing messenger RNA into proteins. By contrast, in mice lacking MNK2, the expression and phosphorylation of proteins responsible for transmitting messages in synapses was reduced.
Notably, the researchers found that the molecular differences between the two kinases were far more pronounced in synapses than in whole-brain samples.
“We were surprised to see these differences,” says Hörnberg. “This suggest that the kinases may have different functions in the cell body compared to synapses.”
Toward more selective therapies
Instead of broadly inhibiting both kinases, future therapies might selectively target one kinase or the other to achieve more specific effects, Hörnberg adds.
She and her colleagues now aim to identify other molecules with which MNK1 and MNK2 interact. They also plan to study in greater detail, how these kinases regulate translation of messenger RNA into proteins during synaptic plasticity – the strengthening or weakening of connections between neurons over time. Synaptic plasticity is the fundamental mechanism underlying learning and memory – enabling the brain to rewire itself based on experience.
By clarifying how protein synthesis is fine-tuned at synapses, the team hopes to lay the groundwork for more precise interventions in neurological and psychiatric disorders.
Text: Gunjan Sinha
Photo: Section of mouse brain tissue showing that messenger RNA for MNK1 (magenta) and MNK2 kinase (turquoise) – important enzymes that regulate the production of proteins – are both expressed in excitatory neurons (green) in the cortex.
© Erika Uddström, Hörnberg Lab, Max Delbrück Center
Source: Press Release Max Delbrück Center
Similar kinases play distinct roles in the brain
Research / 18.02.2026
New material boosts MRI image quality
MRI scans often struggle to capture clear images of deep or delicate tissues. Researchers at the Max Delbrück Center now report in “Advanced Materials” a new lightweight antenna that boosts image quality and may shorten scan times ¬– without changing existing MRI machines.
Magnetic resonance imaging (MRI) is one of medicine’s most powerful diagnostic tools. But certain tissues deep inside the body – including brain regions and delicate structures of the eye and orbit that are of particular relevance for ophthalmology – are difficult to image clearly. The problem is not the scanner itself, but the hardware that sends and receives radio signals.
Now, researchers led by Nandita Saha, a doctoral student in the Experimental Ultrahigh Field Magnetic Resonance lab of Professor Thoralf Niendorf at the Max Delbrück Center have developed an advanced materials-based MRI antenna that overcomes these limitations – delivering enhanced images more quickly and that can be used in existing MRI machines. The research was published in “Advanced Materials.”
Niendorf and his team worked closely with researchers at Rostock University Medical Center, combining expertise in MRI physics with clinical ophthalmology and translational imaging. The Rostock team is also supporting clinical validation of the technology.
“By using concepts from metamaterials, we were able to guide radiofrequency fields more efficiently and demonstrate how advanced physics can directly improve medical imaging,” says Niendorf, senior author of the paper. “This work shows a pathway toward faster, clearer MRI scans that could benefit patients in many clinical areas.”
Rethinking MRI hardware with metamaterials
MRI works by sending radiofrequency (RF) signals into the body and detecting how tissues respond inside a strong magnetic field. The stronger the signal response, the better the image. Conventional MRI antennas – also called RF coils – often struggle to collect enough signal from deep or anatomically complex regions. This leads to images that lack detail and prolongs scan times.
The research team addressed this bottleneck by integrating metamaterials directly into the MRI antenna. Metamaterials are engineered structures that interact with electromagnetic waves in ways not found in natural materials. The engineered RF antenna increases signal strength from targeted tissues, improves spatial resolution and image sharpness and enables faster data acquisition. Crucially, the antenna fits into existing MRI systems, avoiding the need for new infrastructure. The team validated the technology by imaging the eye and orbit region in a group of volunteers at 7.0 Tesla.
"Our research demonstrates clear relevance for ophthalmological applications as it can facilitate anatomically detailed, high-spatial resolution MRI of the eye,” says Professor Oliver Stachs, a co-author of the paper at University Medicine Rostock. “It offers the potential to open a window into the eye and into (patho)physiological processes that in the past have been largely inaccessible.”
“Our goal was to rethink MRI hardware from the modern physics of antenna design,” adds Saha. This technology can also be tuned to protect sensitive areas of the body during MRI, for example, to reduce unwanted heating around medical implants, she adds. It could also be used to focus RF energy more effectively for MRI guided therapies for various cancer treatments, such as gentle heating of tumors (hyperthermia) or thermal ablation of tissue.
Better diagnostics
For patients, MRI scans can be uncomfortable and time-consuming – even more so when images need to be repeated because important details are hard to see. Faster scans mean patients spend less time inside scanners. Clearer images mean doctors can make diagnoses with greater confidence. And because the new antenna is lightweight and compact, it can also be designed to better fit specific parts of the body, improving comfort even further.
The technology could also be adapted to support MRI systems running at magnetic field strengths lower or higher than 7.0 T, to image target anatomy other than the eye, orbit or the brain or to track metabolism or drug movement inside the body, says Niendorf. Special MRI scans that use other atoms, such as sodium or fluorine, could also benefit from this technology by producing clearer signals and better images, he adds.
“Innovations in imaging hardware have the potential to transform diagnostics, and this study is an important step toward next-generation MRI technology,” says Dr. Ebba Beller, a co-author of the paper at Rostock University Medical Center.
The researchers are already planning larger studies at multiple hospitals and adapting the design for other organs, such as the heart and kidneys. The collaboration will continue to be strengthened by long-standing reciprocal visiting scientist appointments of Stachs and Niendorf.
*This project was funded by the DFG as a joint collaboration between the Max Delbrück Center and the Medical University Rostock.
Source: Max Delbrück Center
New material boosts MRI image quality
Research / 06.02.2026
Leif Si-Hun Ludwig awarded professorship

Leif Si-Hun Ludwig has been awarded a prestigious Heisenberg professorship in stem cell dynamics and mitochondrial genomics by the the Berlin Institute of Health at Charité, a position funded by the German Research Foundation (DFG).
Professor Leif Si-Hun Ludwig has led an Emmy Noether Research Group at the Berlin Institute of Health and the Max Delbrück Center since 2020. He has now been awarded a Heisenberg Professorship for Stem Cell Dynamics and Mitochondrial Genomics by the the Berlin Institute of Health (BIH) at Charité, a position funded by the German Research Foundation. Ludwig’s professorship will transition into a permanent role following an initial five-year funding period.
Studying how blood cells form
The human blood system is a marvel of constant regeneration, with millions of new cells replacing old blood and immune cells every second. These cells originate from hematopoietic (or blood-forming) stem cells in the bone marrow, maturing through several different developmental stages into the red and white blood cells, platelets, and B and T cells that keep us alive. While clinical blood tests can easily count these cells, determining the specific contribution of thousands of individual stem cells to overall blood production remains a significant challenge.
By observing natural mutations in human DNA, researchers can gain fundamental insights into how stem cells maintain healthy blood formation or how they behave when disease strikes. However, hunting for a single mutation within a genome of three billion base pairs is expensive, time-consuming and prone to error, even with today’s most advanced technology.
Cellular power plants reveal the origin of blood cells
To bypass these hurdles, Ludwig focuses on natural mutations within the mitochondrial genome – a much smaller, more manageable DNA molecule found within cellular power plants known as mitochondria. By pairing this approach with sophisticated single-cell sequencing technologies, his team can analyze tens of thousands of blood and bone marrow cells simultaneously, effectively mapping the activity of blood-forming stem cells. This single-cell analysis of natural genetic variation does more than track lineage; it provides vital information on the health of individual cells. In a clinical setting, this could eventually help doctors to predict the success of stem cell transplants or fine-tune cell and gene therapies with unprecedented precision.
Beyond this work on hematopoietic stem cells, Ludwig is tackling the challenge of inherited mitochondrial mutations. These defects are among the most common genetic disorders and can trigger a wide range of metabolic diseases affecting multiple organ systems. Despite their prevalence, the molecular causes of these conditions remain poorly understood. By investigating how mitochondrial gene variants influence different cellular and metabolic phenotypes, Ludwig aims to lay the groundwork for new therapeutic approaches.
“The Heisenberg program supports outstanding scientists, so it was no surprise to us that Ludwig was selected for this honor,” says Prof. Christopher Baum, Chair of the BIH Board of Directors and Chief Translational Research Officer of Charité. “His exceptional work combines basic research and application-oriented studies. Ludwig and his team are thus strengthening the translational network that brings together the BIH, Charité, and the Max Delbrück Center for the benefit of patients.”
Research / 06.02.2026
Professorship awarded to Mina Gouti

Mina Gouti has been awarded a professorship at Charité – Universitätsmedizin Berlin. The appointment will strengthen her pioneering organoid research at the Max Delbrück Center and deepen collaboration with clinicians to advance personalized medicine.
Dr. Mina Gouti, Group Leader of the Stem Cell Modeling of Development and Disease lab at the Max Delbrück Center, has been appointed W3 Professor of Complex Organoid Models for Personalized Medicine in the Medical Faculty, Charité – Universitätsmedizin Berlin. The permanent position recognizes her achievements in developing advanced organoid systems to understand how specific types of spinal cord neurons and skeletal muscle cells grow in space and time during development.
“This professorship will enable my team to fully leverage Berlin’s exceptional biomedical ecosystem and to work closely with clinicians at Charité to create complex organoids for personalized medicine,” Gouti says. This close interaction is essential for translating patient-derived organoid models into clinically relevant insights.”
The Gouti lab has developed three-dimensional neuromuscular organoids from human pluripotent stem cells that replicate key features of spinal cord neurons and skeletal muscle – providing powerful platforms for studying neuromuscular diseases such as spinal muscular atrophy and amyotrophic lateral sclerosis, and for drug screening. Such organoids can also be grown from cells derived from individual patients to model their specific disease.
The position reflects a shared vision between the Max Delbrück Center and Charité to accelerate translational research through fostering closer collaboration between clinicians researchers. “We now have the long-term perspective required to develop complex, functional organoids as predictive platforms for personalized medicine and early disease interception,” says Gouti. “We hope our research will lead to better health outcomes not only for people with neuromuscular diseases, but also those at high risk of developing them.”
Text: Gunjan Sinha
Further information
Research, Innovation / 05.02.2026
Pentixapharm publishes positive Phase II data on Pentixafor PET diagnostics
The study confirms PENTIXAFOR-PET as a non-invasive alternative to adrenal vein catheterization in primary aldosteronism
-
In the study, [68Ga]Ga-Pentixafor PET/CT was well tolerated and demonstrated high specificity and moderate sensitivity for identifying unilateral aldosterone-producing adenomas compared with adrenal vein sampling (AVS) and surgical outcomes
-
The data strengthens the clinical foundation for Phase 3 development and support the role of molecular imaging in guiding treatment decisions in the population with hypertension and underlying primary aldosteronism
Pentixapharm Holding AG (Frankfurt Prime Standard: PTP), an advanced clinical-stage biotech developing novel radiopharmaceuticals, today announced publication of new Phase 2 clinical data in the Journal of Nuclear Medicine demonstrating the potential of [68Ga]Ga-Pentixafor PET/CT as a non-invasive imaging tool for subtyping primary aldosteronism (PA), the leading endocrine cause of hypertension.
The investigator-initiated study, funded by an Australian philanthropic foundation, the CASS Foundation, and the Medical Research Future Fund, was conducted as a prospective cohort study in Australia. It evaluated [68Ga]Ga-Pentixafor PET/CT as a potential alternative to adrenal vein sampling (AVS), the current standard of care for distinguishing unilateral aldosterone-producing adrenal adenomas from bilateral disease. AVS is invasive, resource-intensive, and available only in highly specialized centers, creating barriers to timely and accurate patient stratification.
Results published online show that [68Ga]Ga-Pentixafor PET/CT demonstrated high specificity and moderate sensitivity for identifying unilateral aldosterone-producing adenomas when compared with AVS and surgical outcomes. Importantly, the imaging approach was well tolerated and strongly preferred by patients, with 28 of 29 participants indicating PET/CT as the favoured diagnostic test.
“These data provide evidence that molecular imaging with [68Ga]Ga-Pentixafor PET/CT can support accurate and patient-friendly subtyping of primary aldosteronism,” said Dr. Elisabeth Ng, of the Hudson Institute of Medical Research and the Endocrinology Unit at Monash Health, the lead investigator of the study. “The high specificity observed is particularly relevant for identifying patients who may benefit from curative surgery, while the strong patient preference for PET/CT highlights the potential to expand access to and acceptance of diagnostic testing for patients with primary aldosteronism.”
The study recruited adults with primary aldosteronism and an adrenal adenoma visible on CT imaging. Diagnostic performance was assessed by comparing PET-derived lateralisation indices with AVS results and biochemical outcomes following adrenalectomy. The findings support the clinical utility of [68Ga]Ga-Pentixafor PET/CT as a noninvasive decision-support tool for identifying patients who may benefit from curative surgery.
“This published data builds on earlier investigator-initiated studies, including the CASTUS Step 1 trial and demonstrates reproducible performance across independent studies and geographies. Together, these data strengthen the clinical foundation of Pentixapharm’s primary aldosteronism program and support readiness for Phase 3 development,” said Dirk Pleimes, CEO and CMO of Pentixapharm. “Here, at Pentixapharm, we are continuing to advance our clinical and regulatory strategy for primary aldosteronism while engaging with investigators, regulators, and potential partners to maximise the clinical and commercial impact of our molecular imaging platform.”
As new therapeutic options, including aldosterone synthase inhibitors, are expected to enter the treatment resistant hypertension market, accurate and scalable subtyping of primary aldosteronism is becoming increasingly important. Noninvasive imaging solutions may play a critical role in guiding treatment decisions and optimising patient outcomes in this evolving therapeutic landscape.
The full article, titled “Identification of Aldosterone-Producing Adrenal Adenomas Using [68Ga]Ga-Pentixafor PET/CT in an Australian Cohort,” is available in the Journal of Nuclear Medicine.
About 68Ga-PentixaFor in treatment-resistant hypertension and primary aldosteronism
[68Ga]Ga-PentixaFor is a novel gallium-68-labeled radiodiagnostic designed to selectively target and visualize the chemokine receptor CXCR4 using high-resolution PET/CT imaging. Clinical experience with [⁶⁸Ga]Ga-PentixaFor PET/CT in approximately 1,600 patients across different indications has demonstrated its ability to non-invasively image CXCR4 expression in vivo.
Recent research has shown strong CXCR4 overexpression in aldosterone-producing adrenal tumors, a hallmark of unilateral primary aldosteronism. Primary aldosteronism is a common but historically underdiagnosed cause of secondary hypertension, largely because reliably distinguishing unilateral from bilateral disease remains challenging with current diagnostic tools. Unilateral disease is typically treated by surgical removal of the affected adrenal gland whereas bilateral disease requires life-long medical therapy. By visualizing CXCR4 expression in aldosterone-producing tissue, [⁶⁸Ga]Ga-PentixaFor has the potential to support more reliable subtyping of primary aldosteronism and thereby better guide appropriate treatment decisions.
About the prospective phase 2 pilot study
The prospective pilot study recruited adults with PA and an adrenal adenoma visible on CT and evaluated 68Ga-Pentixafor PET/CT as a noninvasive nuclear imaging alternative to AVS, assessing its diagnostic accuracy and acceptability compared with AVS in a multiethnic population. PentixaFor was supplied by Pentixapharm AG. The study was published in the Journal of Nuclear Medicine (JNM), which is a top-ranked peer-reviewed publication covering molecular imaging, PET/CT, and theranostics [Ng E, Jong I, Lau KK, Akram M, Morgan J, Nelva P, Simpson I, Haskali MB, Fuller PJ, Shen J, Yang J. Identification of Aldosterone-Producing Adrenal Adenomas Using [68Ga]Ga-Pentixafor PET/CT in an Australian Cohort. J Nucl Med. 2026 Jan 29:jnumed.125.271006. (doi: 10.2967/jnumed.125.271006. Epub ahead of print. PMID: 41611475].
About Pentixapharm
Pentixapharm is an advanced clinical-stage biotech expanding the boundaries of radiopharmaceuticals. Headquartered in Berlin, Germany, the company develops precision diagnostics and therapeutics in oncology and cardiology to transform patient care. Its clinical pipeline is anchored by CXCR4-targeted PET-CT programs, including a Phase 3-ready candidate for the improved diagnosis of hypertensive patients with primary aldosteronism, which is intended to enable targeted treatment of the underlying causes of hypertension. CXCR4-based developments also include pioneering therapeutic programs in hematological cancers. Furthermore, Pentixapharm is advancing a next-generation antibody platform targeting CD24, an emerging immune-checkpoint marker over-expressed in multiple hard-to-treat cancers. Complemented by CXCR4 and CD24 intellectual property protection and a reliable isotope supply chain, Pentixapharm is poised to deliver meaningful patient benefit and sustainable growth in one of the fastest-growing areas of precision medicine.
Innovation / 29.01.2026
Eckert & Ziegler on Track as Planned and Achieves Another Record Year in 2025
Fiscal Year 2025 (preliminary):
- Sales: approx. € 312 million (PY: € 295.8 million)
- EBIT before special items: approx. € 78 million (PY: € 65.9 million)
- Net income: approx. € 48 million (PY: € 33.3 million)
According to preliminary, unaudited figures for the 2025 financial year, Eckert & Ziegler SE (ISIN DE0005659700, TecDAX) generated sales of around € 312 million and adjusted EBIT of around € 78 million. Sales are up around 5% on the previous year, while adjusted EBIT is up by around 18%. Net profit (from continuing and discontinued operations), which is only reported here for comparison purposes, rose to around € 48 million (previous year: € 33.3 million).
The forecast for the 2026 financial year will be published on 26 March 2026 together with the complete, audited annual financial statements for the 2025 financial year.
About Eckert & Ziegler.
Eckert & Ziegler SE with more than 1.000 employees is a leading specialist for isotope-related components in nuclear medicine and radiation therapy. The company offers a broad range of services and products for the radiopharmaceutical industry, from early development work to contract manufacturing and distribution. Eckert & Ziegler shares (ISIN DE0005659700) are listed in the TecDAX index of Deutsche Börse.
Contributing to saving lives.
economic development / 29.01.2026
OMEICOS Therapeutics Announces Positive Phase 2 Study Outcome Demonstrating OMT-28’s Potential in Primary Mitochondrial Diseases (PMD)
Trial Results Support Transition into Late-Stage Development with Program Expected to be Phase 2b/3-ready in H2 2026
OMEICOS, a clinical-stage biopharmaceutical company developing first-in-class small molecule therapeutics for mitochondrial and inflammatory disorders, announced the successful conclusion of its multi-centre, open-label Phase 2a PMD-OPTION Study evaluating its lead program OMT-28 in patients with Primary Mitochondrial Disease (PMD). The study results demonstrate OMT-28’s therapeutic potential to improve the physical condition in PMD based on significant recovery of the impaired mitochondrial fitness in the responding patients. The study further underscored the excellent safety and tolerability profile of OMT-28, which has now been evaluated in more than 220 individuals. OMEICOS is preparing for a potentially pivotal Phase 2b/3 study in PMD patients with myopathy and/or cardiomyopathy across EU and US sites and expects to be ready to initiate this study later this year, subject to the completion of partnering discussions.
PMD represents a heterogeneous group of conditions including the more prevalent subtypes MELAS, non-MELAS, and MIDD. PMD patients suffer from debilitating and life-threatening health consequences, such as severely limited physical stamina and disease-related changes in the heart and skeletal muscles, as well as associated neurological disorders. OMT-28, an orally available biased modulator, targets the GPCR-receptor S1PR1 (Sphingosine-1-Phosphate Receptor 1 thereby driving downstream activation of the mitochondrial sirtuin family members SIRT1 and SIRT3. By targeting S1PR1 and activating SIRT1/SIRT3, OMT-28 combines immunomodulation with mitochondrial protection—a dual mechanism to tackle inflammation and energy deficits in primary mitochondrial diseases.
“Improving physical performance through enhanced mitochondrial metabolism and reduced oxidative stress holds great promise in PMD. Our PMD-OPTION study results indicate a strong correlation between OMT-28 treatment, the observed positive impact on mitochondrial bioenergetics and fitness, and relevant clinical improvements in functional measures, which could translate into significant patient benefit,” said Dr. Robert Fischer, CEO/CSO of OMEICOS Therapeutics. “The profound effects on NAD⁺ and GSH levels, as well as simultaneous improvement of the NAD⁺/NADH and GSH/GSSG ratios we have seen in the responder group, are integrative indicators of electron transport chain function improvements and cellular redox homeostasis. Overall, the results offer a robust path for late-stage development.”
Study Design and Results Summary
The PMD-OPTION study enrolled a total of 29 PMD patients with mitochondrial tRNA point mutations or single mtDNA deletions across nine expert sites in Germany, Italy, and The Netherlands. The study generated strong interest among patients and key opinion leaders (KOLs), resulting in timely recruitment and a high degree of compliance with the study protocol and follow-up appointments. The study included a 12-week untreated run-in phase as an integrated control, capturing the patients’ natural history and baseline parameters for evaluating treatment results. Subsequently, all patients received a 24 mg once-daily dose of OMT-28 for a treatment period of up to 24 weeks. The study ended after a subsequent four-week follow-up period. The level of GDF-15, a prospective biomarker for reflecting cellular stress and inflammation, was used as a screening and inclusion criteria, while reduction of GDF-15 was used as a primary endpoint next to demonstrating safety and tolerability in PMD patients. The study outcome did not support the choice of GDF-15 in this setting suggesting that OMT-28 is acting downstream of the release mechanism of GDF-15.
To assess clinically meaningful improvements in the study population, the PMD-OPTION study utilized a combination of objective exercise endpoints and patient-reported outcomes. Using these measures, the study demonstrated a response rate of more than 60%. In the 12-Minute Walk Test (12 MWT) and the 5x Sit-to-Stand Test (5xSST), both accepted endpoints for pivotal studies, the entire study population showed improvements over baseline, while OMT-28 responders exhibited profound and statistically significant (12 MWT) clinical improvements compared to non-responders.
These results strongly correlated with a highly significant increase in total NAD+ levels in the responder group compared to baseline, and a clear separation between responders and non-responders in NAD+/NADH ratios over the course of the study. In patients responding to OMT-28 treatment, mean NAD+ levels were approximately 30% higher compared to baseline, bringing this crucial indicator of mitochondrial energy metabolism and redox status close to healthy ranges. Similarly, OMT-28 demonstrated a significant improvement in total GSH and GSH/GSSG ratios—key indicators of reduced oxidative stress in mitochondrial diseases—thereby reestablishing normal, healthy levels and even showing a trend toward further enhancement. Together, these results demonstrate that OMT-28’s ability to normalize both NAD⁺/NADH and GSH/GSSG ratios addresses the core pathologies of PMD—energy deficiency and oxidative stress—differentiating it from single-mechanism approaches and supporting its potential as a first-in-class therapy.
About OMEICOS
OMEICOS Therapeutics has discovered a series of metabolically robust synthetic analogues of omega-3 fatty acid-derived epoxyeicosanoids that have the potential to treat mitochondrial dysfunction, inflammatory, cardiovascular and other diseases. Epoxyeicosanoids activate cell type-specific endogenous pathways that promote organ and tissue protection. OMEICOS’ small molecules are orally available and show improved biological activity and pharmacokinetic properties compared to their natural counterparts. For more, please visit: www.omeicos.com
Innovation / 14.01.2026
A fresh take on proteomics

Start-up company Absea Biotechnology GmbH is developing new proteomics technologies. An interview with Dr. Philip Lössl, Senior VP Science and Business Development
What makes the Absea Biotechnology Group special?
Absea Biotechnology develops protein science technologies to advance proteomics. One of our main areas of focus is developing monoclonal antibodies for the entire human proteome. These antibodies can detect highly specific molecular biomarkers or diseased cells, such as those associated with cancer or autoimmune diseases. In collaboration with our sister companies in China and the U.S., we have amassed the world’s largest library of recombinant human proteins, which we are continually expanding through our high-throughput protein production platform. Alongside this, we are establishing mass spectrometry pipelines in Berlin to better understand protein–drug interactions (PDIs).
We see ourselves as partners and service providers in proteomics, in vitro diagnostics, and pharmaceutical and life sciences research.
How did the Absea Biotechnology Group come about?
Immunology professor Wei Zhang laid the groundwork. While earning her Ph.D. at Cambridge, she collaborated with Nobel Prize winners César Milstein and Gregory Winter, who studied the fundamentals of monoclonal antibodies. After founding her first antibody company, Zhang worked closely with the Human Protein Atlas project. Absea later developed an even larger partnership with Olink, a Swedish company that is now part of ThermoFisher. Since then, Absea has developed thousands of protein antigens and monoclonal antibodies for Olink. In 2020, the Absea Group was reorganized around bioinformatician Tao Chen to better support this proteome-wide approach. In 2023, we established our Berlin-based start-up, focusing on mass spectrometry research and development.
What is the secret behind Absea’s extensive protein library?
Tao Chen developed a machine learning-based algorithm for designing protein constructs. These constructs correspond to natural sequences found in our bodies, though they are sometimes slightly shorter. Thanks to the algorithm, we know exactly how to shorten the proteins in order to produce them efficiently and cost-effectively with a high success rate.
What innovations are you pursuing?
Our antibody and protein libraries map the proteome and support affinity-based methods. This treasure trove of molecules can also help us develop more unique mass spectrometry technologies and complement “making molecules” with “mapping molecules.” Our scientific advisors, Mikhail Savitski from the European Molecular Biology Laboratory (EMBL) in Heidelberg and Fan Liu from the Leibniz-Forschungsinstitut für Molekulare Pharmakologie (FMP), both leading experts in mass spectrometry proteomics, encouraged us in this endeavor. Together with Matthias Mann– Germany’s most cited researcher, one of the founding fathers of proteomics, and a researcher at the Max Planck Institute of Biochemistry (MPIB) in Martinsried – we published an approach to using our proteins for clinical testing mass spectrometry.
What does this approach entail?
Proteomics typically only allows for the relative quantification of protein biomarkers: a physician could tell you that you have more of a certain protein in your blood than last time, but they could not tell you the exact amount. Therefore, absolute quantification of biomarkers is important for clinical diagnostics. Until now, peptides labeled with heavy isotopes have been used for this purpose. However, this method only allows the part of the protein biomarker covered by the peptide to be seen and quantified. For this reason, we started producing whole proteins labeled with isotopes. Until now, this was not possible on a large scale. Thanks to our many years of experience in protein production, however, we have figured out how to do this.
When these labeled proteins are added to the sample, they are much more similar to the biomarker because both are proteins. Unlike peptides, proteins can be included in sample preparation. This enables researchers to monitor where deviations occur. During sample preparation, the biomarker and the labeled protein are broken down into peptides. The isotope-labeled counterpart is also found in the peptides. This provides a large number of data points for quantifying the biomarker. If the correlation with the labeled counterpart is incorrect, further mass spectrometry measurements can determine what happened in this region.
This allows parallel and highly sensitive testing of entire protein panels for a specific disease to detect abnormal quantities. At this level, the results could be relevant to clinical practice.
A path to clinical application would therefore be possible.
That is our hope. The plasma proteomics community is proposing the same approach. We are already working with a European university hospital, but we are still in the research and development phase.
However, we have even more plans: we are developing a methodology platform to better map protein–protein and protein–drug interactions at the molecular level. Our goal is to analyze these interactions directly in intact cells, tissue lysates, and cell lysates to gain a better understanding of diseases and the effects of drugs. Ultimately, we want to help our partners develop more effective drugs.
How would you go about doing that?
The first step is to determine if a drug binds to the intended protein and if there are any other unexpected – or even undesirable – binding partners. We use mass spectrometry technology to accomplish this. This technology can show us which protein areas are blocked by a drug across the entire proteome. We also discover the precise location where the drug binds. This information tells us, for example, whether the drug can inactivate the protein or prevent it from interacting with other proteins.
The second step involves protein-protein interactions (PPIs). Which PPIs occur in treated or untreated cells? Which ones occur in healthy or diseased cells? Crosslinking mass spectrometry provides an overview of the entire cellular network. Using our antibody libraries, we can then go into greater depth: we can use crosslinking to fix all protein–protein contacts in the cell. Then, the protein of interest can be enriched using one of our numerous antibodies and extracted from the cell with all its interaction partners for analysis.
Which customers are you targeting with this approach?
For pharmaceutical customers, we can identify disease-relevant protein targets for drugs in the early stages of development. By showing exactly how drugs work, we can determine which candidates are worth further development.
Another customer group consists of protein biotechnology companies that specialize in AI. Our comprehensive, high-quality datasets contain thousands of PPIs and PDIs, providing these companies with valuable information for developing their AI models.
Starting in January, your start-up will be located in the BerlinBioCube incubator
We look forward to being in the same building as the other start-ups, exchanging ideas and supporting each other. I believe this is an ideal ecosystem for us to grow in. We already have academic collaborations on campus with the FMP and the Max Delbrück Center. We also have strong relationships with biotech companies. We see a lot of potential for joint projects at this location.
The interview first appeared in Standortjournal buchinside 1/2026.
Research / 09.01.2026
A double-pronged attack on malignant B cells
Multiple myeloma, a cancer of the bone marrow, remains difficult to treat despite modern CAR T cell therapies. In “Molecular Therapy,” a team led by Armin Rehm presents an improved immunotherapy that recognizes two distinct features of malignant cells to destroy the cancer more effectively.
Multiple myeloma is a devastating diagnosis. The disease usually develops in the bone marrow where mature B cells called plasma cells begin to proliferate uncontrollably and produce excessive amounts of antibodies, some of which are defective. The cancer, which destroys bone and other tissues, can become extremely painful. To date, it remains uncurable.
In recent years, modern CAR T cell therapies have significantly extended the lives of many myeloma patients, but their effectiveness has been limited. “In some patients, the treatment does not work at all. In others, relapse occurs sooner or later, which then often rapidly leads to death,” explains Dr. Armin Rehm, Group Leader of the Translational Tumorimmunology lab at the Max Delbrück Center.
In the journal “Molecular Therapy,” the Berlin-based researcher, Dr. Uta Elisabeth Höpken, also at the Max Delbrück Center, and an international team present an optimized immunotherapy: CAR T cells that bind to two receptors simultaneously to destroy cancer cells instead of just one, as was previously the case.
Not all patients express the BCMA receptor
Before the advent of CAR T cell therapy, multiple myeloma was treated with chemotherapy and antibody-based drugs, and sometimes combined with autologous stem cell transplants. These approaches, however, were not curative either. CAR T-cell therapies gave patients new hope.
The treatment involves collecting T cells from patients and genetically engineering them in a bioreactor to express a chimeric antigen receptor, or CAR. These receptors act as sensors that help immune cells detect specific surface proteins. In the case of multiple myeloma, the target is BCMA, a protein found almost exclusively on plasma cells, and not on normal B cells. The modified T cells are then reinfused into patients, where they seek out and destroy cancer cells in the body.
“However, BCMA protein isn’t present on all malignant plasma cells,” says Rehm. In some patients with multiple myeloma, it’s not detectable at all; in others, it disappears over the course of treatment. “That’s why we combined three methods – single-cell RNA sequencing, immunohistochemistry, and flow cytometry – to search for another surface marker characteristic of malignant B cells. That led us to BAFF-R,” says Dr. Agnese Fiori, the study’s first author and a member of Rehm’s team, who now conducts research at Charité- Universitätsmedizin Berlin.
Prevention and treatment of relapse
Two additional researchers also made significant contributions to the study: Professor Jörg Westermann, Deputy Clinic Director and Head of Hematological Diagnostics at Charité, and Professor Jan Krönke, Head of the Department of Internal Medicine C – Hematology, Oncology, Stem Cell Transplantation, and Palliative Medicine at Greifswald University Medical School.
“BAFF-R is particularly abundant on cells from multiple myeloma patients who have relapsed,” says Fiori. In addition, it is often found on abnormal plasma cells without BCMA, which are resistant to conventional treatments. “Therapeutically, it therefore makes sense in two respects to use CAR-T cells that recognize both BCMA and BAFF-R,” explains Rehm.
Around 5 to 30 percent of all patients with multiple myeloma could benefit from this dual approach, adds Rehm. For personalized therapy, it is crucial to identify these patients using an appropriate test before treatment begins, he explains. Such screening would be easy to implement in clinical practice.
Success in experiments with cancer cells
Rehm and his colleagues have tested the bispecific CAR T cells both in multiple myeloma cell lines and in patient-derived bone marrow cells. “In all of our experiments, we showed that the improved CAR T cells remain effective, even when the BCMA receptor is absent or has been lost due to therapy – that is, even when treatment with monospecific CAR T cells has failed,” he explains. He hopes that the new approach can prevent relapse in many patients and also extend lifespan – perhaps even offer a cure.
“To truly assess the potential of our method, an initial clinical trial is now necessary,” Rehm adds. Together with colleagues from the National Center for Tumor Diseases (NCT) – a long-term collaboration between the German Cancer Research Center (DKFZ), excellent partners in university medicine, and other outstanding research partners at various locations in Germany, including the Max Delbrück Center, Charité, and the Berlin Institute of Health (BIH) as partner sites in Berlin – he plans to apply for the requisite funding. He hopes patients may soon be able to benefit from the optimized CAR T cells.
Text: Anke Brodmerkel
www.mdc-berlin.deResearch / 08.01.2026
A new opportunity for Berlin

The Einstein Center for Early Disease Interception has been funded with six million euros. In an interview, Nikolaus Rajewsky and Leif Erik Sander, both spokespeople of the center, explain why new technologies and cross-institutional collaboration will make disease prevention in the future possible.
The phrase “early disease interception” is included in the Einstein Center’s name. What does that mean in concrete terms?
Professor Nikolaus Rajewsky: Pathological processes in tissues often progress for years unnoticed before symptoms appear. By that point, the damage is usually substantial and can only be reversed to a limited extent. It would be far better to intervene much earlier at a stage when only individual cells are affected and when we can better influence the disease trajectory.
Thanks to major technological advances, we now effectively have a molecular super microscope. We can make disease processes visible at subcellular resolution – in tissues, in body fluids, even in the air we exhale. Artificial intelligence and machine learning are helping us to manage the enormous volumes of data generated from our research and predict disease trajectories.
We want to understand the earliest mechanisms that drive cells away from a healthy equilibrium toward disease and to control which path these cells take – that is what we mean by “interception.” We can now realistically test such targeted interventions before damage occurs, for example using organoids, which are miniaturized models of organs.
Professor Sander, how would you explain this approach to your patients?
Professor Leif Erik Sander: At its core, our approach is about very precise prevention. What matters to patients is that their quality of life and organ function are preserved – even if pathological changes have already begun at the cellular level.
In cancer screening, such approaches already exist, albeit using more conventional methods. For many other diseases, however, we currently intervene too late. When dementia is diagnosed, for example, we can only slow progression of the disease. A similar situation exists with certain lung diseases that can cause progressive tissue scarring, which can lead to death.
Conventional methods cannot detect the early warning signs of such diseases – and we do not always understand what is actually going wrong or how to intervene. This is where early disease interception comes in: we want to understand what knocks the system off course, and create opportunities to reset it at very early stages. The train should, so to speak, be put back on a healthy track.
How exactly does this super-microscope work?
Nikolaus Rajewsky: Today we can analyze very precisely at what point in time, which cells in a tissue access which information in their genome – or do not. A single histological section contains around 100,000 cells, each with about 20,000 genes. Accordingly, the resulting data volumes are enormous. Based on these data, we can reconstruct disease-relevant signaling pathways and complex metabolic networks. The decisive advance is that we no longer merely describe disease, but understand it causally. This is the prerequisite for targeted and early intervention.
What are the biggest hurdles on the path to clinical application?
Leif Erik Sander: These extremely high-resolution methods provide exactly the information we need to distinguish early disease processes. Right now, they are research tools and are very expensive. The crucial next step will be to greatly reduce the complexity and to develop robust, simplified tests that are suitable for large numbers of people.
Artificial intelligence will play a key role. We want to use it to link molecular data with routine clinical data, identify patterns, and specifically predict individual disease risks.
The Einstein Center will be funded for six years. What do you expect to achieve during this time?
Nikolaus Rajewsky: What is emerging here in Berlin goes beyond individual projects. Together, we are driving forward a new form of molecular prevention. We developed this concept within the European LifeTime consortium and described it in the journal “Nature” in 2020. Hundreds of scientists from across Europe were involved, so we are extremely well connected internationally.
With the Berlin excellence cluster ImmunoPreCept, there is a strong complementary focus on immunology. What is new with the Einstein Center is a structured, cross-institutional collaboration to find new paths toward application. Clinicians, basic researchers, and data scientists will work together systematically with their respective expertise, rather than alongside one another.
Major advances arise neither solely in the clinic nor exclusively in the laboratory nor at the computer. The Einstein Center will create a shared platform with clear rules, short paths, and without unnecessary bureaucratic hurdles. This is absolutely essential because complex collaborations otherwise often fail before they even begin.
During the two-year preparatory phase for the Einstein Center, we looked internationally for role models. There is an interesting structure in Lausanne. Then there is the Broad Institute in Boston, where Harvard, MIT, and others have joined forces. In Berlin, we want to gradually create an open network in which the basic rules are defined for everyone interested, making processes faster as a result. By the way, the topic of molecular prevention is also being addressed elsewhere in Germany and internationally.
Is Berlin well positioned to advance this concept?
Leif Erik Sander: We have state-run universities that provide education at a very high level. With Charité, we have the largest university hospital in Europe, as well as a very high density of non-university research institutions where top minds work with the most modern technologies. They contribute the expertise required for such a complex and forward-looking project. For me, this combination is Berlin’s competitive advantage that will quickly result in real added value.
For highly qualified young talent in Berlin, institutional boundaries hardly matter. They have great ideas and want to freely use technologies from different institutions to make new discoveries. That is why they come here. The Einstein Center will provide a structural framework for this. Of course, the next Broad Institute will not emerge here in just a few years. But something that is driven by a similar spirit will: top institutions working together to make a difference. Collaboration is orders of magnitude better than everyone trying on their own and focusing only on themselves. This will also lead to shared value creation.
Over the next six years, we want to initiate the first joint innovations, ranging from new diagnostic approaches to patents and spin-offs. A key milestone will be the formalized collaboration of Berlin’s leading organizations. This will offer a platform for the future from which a great deal of good can emerge, benefiting not only medicine but also Berlin as an innovation hub.
The project is quite ambitious. Where will you start?
Nikolaus Rajewsky: During the preparatory phase, young researchers came together across institutions and jointly defined initial use cases. This bottom-up dynamic is an important part of our concept.
What are these use cases?
Leif Erik Sander: We are initially focusing on two organ systems for which there is a high medical need. One focus is dementias. From a healthcare cost perspective, they are expensive to manage and are extremely burdensome for people affected and their families. In several interdisciplinary projects, we are searching for new approaches to diagnose dementias as early as possible in order to influence disease progression. The second focus is chronic lung diseases, which are also widespread conditions. It is often unclear how they develop. With current therapy, we try to slow their progression. But it would be far better to detect these diseases much earlier and cure them.
In addition, we are investigating how organ systems communicate with one another. Damage in one organ can trigger domino effects elsewhere in the body. Such systemic connections have not been not readily apparent within the disease silos in which we have been working; we are now bringing them together. The first two focus areas will hopefully inspire other researchers to use the Einstein Center platform.
Why should people seek medical care even if they feel healthy?
Nikolaus Rajewsky: Many already do, for example through mammography, dental check-ups, or in the case of known genetic risks. This form of prevention will continue to evolve. It is important to conduct societal debate on the issue of prevention openly and transparently.
Leif Erik Sander: If we want to preserve our health and social systems in Germany with an aging population, prevention is indispensable. Leaving it unaddressed would be one of the greatest strategic mistakes one could make. Oral care is a great example. Dentists hardly ever see truly poor dental health anymore. People go for check-ups and professional cleanings, even though it is not always pleasant and sometimes has to be paid for out of pocket. But it pays off in the long term.
The old saying “there is no glory in prevention” is wrong. Only if we intervene earlier and achieve cures instead of chronic long-term therapies will medical progress remain financially sustainable.
What is needed to bring us closer to this goal?
Leif Erik Sander: There is a lack of sufficient venture capital, as is the case everywhere in Europe. Political and tax frameworks also need to be improved. And we do not yet have a strong culture of commercializing our discoveries. Without these things, knowledge does not reach the patient’s bedside! Discoveries lead to medicines, tests, new devices, and eventually they become part of standard care that is covered by medical insurance. That is how medicine works.
Value creation is also part of the Einstein Center’s mission, because profits can then be reinvested. This will generate more cutting-edge research and ultimately create a highly qualified workforce with and well-paying jobs. Within the Einstein Center, we can demonstrate in a protected setting how joint innovation can succeed. This can serve as a blueprint, because in my view, Berlin still has enormous untapped economic potential in the biotech and life sciences sector.
It is an opportunity …
Nikolaus Rajewsky: I was very positively surprised by the enthusiasm with which top scientists in Berlin were involved from the very beginning. This shows that we struck a nerve.
This city needs new ideas for how we want to generate value in the future. Until World War II, there was a lot of industry here. Then mainly administrative structures followed. In recent years, a great start-up scene has emerged. But where Berlin is truly excellent is in biotech and top-level medicine. This is where value creation that benefits everyone can emerge. We are appealing to policymakers in Berlin and at the federal level to seize this opportunity.
In Boston, a huge ecosystem has developed around the biotech industry that contributes to the prosperity of the entire region. There, policymakers spoke to biologists and asked what they needed. The answer was: framework conditions that allow rapid progress. They implemented them. The Einstein Center is a beginning, and we are very grateful to the foundation for its funding. It is an invitation to consider whether such a vision for the future can make Berlin sustainably attractive.
Interview: Jana Schlütter.
Professor Nikolaus Rajewsky is Director of the Berlin Institute for Medical Systems Biology of the Max Delbrück Center. Professor Leif Erik Sander is Medical Director of the Department of Infectious Diseases and Critical Care Medicine at Charité – Universitätsmedizin Berlin. Both are spokespeople for the Einstein Center for Early Disease Interception.
www.mdc-berlin.deResearch, Innovation / 05.01.2026
A spin-off targeting neuromuscular disorders

A team led by Mina Gouti has developed a stem cell-based model that enables researchers to test new drugs to treat neuromuscular diseases and to refine existing therapies. The project, called NMJCare, was named one of the top ten ideas in the national Science4Life start-up competition.
Spinal muscular atrophy (SMA) is one of the most severe genetically inherited neuromuscular disorders, affecting about one in 10,000 children. Despite therapeutic advances in recent years, the condition remains incurable. Researchers continue to rely on reliable, patient-specific models to test new treatments.
Dr. Mina Gouti, head of the Stem Cell Modeling of Development and Disease lab and her team at the Max Delbrück Center have developed just such a model. They have grown structures from patient-derived stem cells that closely mimic the functional interaction between nerve fibers, motor neurons, and muscle fibers in the human body. Dr. Ines Lahmann, a postdoctoral researcher in Gouti’s lab, now plans to transform this scientific innovation into a market-ready product. Her project, NMJCare, has been chosen as one of the ten best submissions out of more than 120 entries in the national Science4Life start-up competition.
The award gives Lahmann access to expert guidance on building a viable business model. NMJCare will also receive support through the GeneNovate Entrepreneurship Program, which offers strategic coaching in company formation, communication, and business planning. “These are all key building blocks for driving targeted technology transfer,” says Lahmann.
One model, many options
NMJCare marks the beginning of a new phase for Lahmann. In addition to scientific questions, her focus is now shifting to topics like patents, target audience analysis, and funding strategies. “Those are things I hardly had to think about before,” Lahmann says. “But now they determine whether our idea can truly become market-ready.” The potential of NMJCare is significant: the model allows high-throughput testing of thousands of potential drug compounds before moving into costly preclinical or clinical trials.
At the same time, the system makes it possible to better understand why patients respond differently to therapies. “That’s a crucial step toward more individualized treatment approaches,” Lahmann explains. Potential users of the model include pharmaceutical companies, academic research groups, and firms in the field of personalized medicine. NMJCare can be flexibly adapted to meet their needs – by selecting specific stem cell lines or investigating short-or long-term drug effects, for example.
“For an idea to become viable, it takes constant exchange – and also critical perspectives from the outside,” says Lahmann. Through the GeneNovate and Science4Life programs, she will be connecting with like-minded peers as well as mentors from business, law, and finance. With their support, she hopes that NMJCare will evolve from a lab-based project into a full-fledged spin-off.
Source: Max Delbrück Center
A spin-off targeting neuromuscular disorders
Research / 19.12.2025
What determines the fate of a T cell?
Researchers at the Max Delbrück Center have found that a cellular housekeeping mechanism called autophagy plays a major role in ensuring that T stem cells undergo normal cell division. The findings, published in “Nature Cell Biology,” could help boost vaccine response in older adults.
When killer T cells of our immune system divide, they normally undergo asymmetric cell division (ACD): Each daughter cell inherits different cellular components, which drive the cells toward divergent fates – one cell becomes a short-lived fighter called an effector T cell, the other cell becomes a long-lived memory T cell.
Research by Professor Mariana Borsa at the University of Oxford and colleagues in the Cell Biology of Immunity lab of Professor Katja Simon at the Max Delbrück Center has shown for the first time that cellular autophagy – a “housekeeping” mechanism by which cells degrade and recycle cell cargo – plays a critical role in this decision process.
“Our study provides the first causal evidence that autophagy plays a central role in ensuring that T cells go through ACD normally,” says Borsa, first author of the paper who now leads a research group at the University of Basel. “We found that when T stem cells divide, daughter cells inherit different mitochondria, which influences the T-cell’s destiny,” says Borsa. “By understanding this process, we can start to think about ways to intervene to preserve the function of immune memory cells as we age.”
Split personality
To study ACD in greater detail, the researchers used a novel “MitoSnap” mouse model in which they could tag mitochondria sequentially and discriminate between those in mother and daughter cells. T-cells contain many mitochondria. By tracking how old, damaged mitochondria were distributed between daughter cells, they found that in healthy cells, autophagy was crucial in ensuring that one daughter cell was clear of old mitochondria. This inheritance profile sent the cell down the path toward becoming a long-lived memory precursor cell – immune cells that “remember” a pathogen and begin rapidly dividing when the pathogen is encountered again. Meanwhile, the other daughter cell that took on the older mitochondria became a short-lived effector T-cell – a type of immune cell that rapidly divides to fight off immediate threat. These cells die when the threat is cleared.
When autophagy was disrupted, however, this careful sorting broke down. Both daughter cells inherited damaged mitochondria and hence, were destined to become short-lived cells.
“It was surprising to see that autophagy plays a role beyond just cellular housekeeping,” says Borsa. “Our findings suggest asymmetric inheritance of mitochondria as a potential therapeutic target for memory T cell rejuvenation.”
Boosting vaccine response
By boosting autophagy before or during T stem cell division, it may be possible to enhance the generation of memory cells – the backbone of long-term immunity and vaccine effectiveness.
What’s more, the researchers analyzed daughter cells using single-cell transcriptomics, proteomics and metabolomics and found that effector cells burdened with damaged mitochondria depend heavily on a metabolic pathway called one-carbon metabolism. Targeting this pathway could offer another way to subtly shift the immune balance – nudging T stem-cells toward becoming memory instead of effector cells, Borsa says.
“In the long run, this research could inform strategies to rejuvenate the aging immune system, making vaccines more effective and strengthening protection against infections,” adds Simon. The researchers are planning to further validate their findings in human T-cells.
Photo: T stem cells normally undergo asymmetric cell division (left) whereby one daughter cell becomes a long-lived memory T cell. When autophagy is disrupted, both daughter cells inherit old mitochondria (red) and become effector T-cells. © University of Oxford
Text: Gunjan Sinha
Source: Press Release Max Delbrück Center
https://www.mdc-berlin.de/news/press/what-determines-fate-t-cell
- News
- Events
- MAGAZINE „buchinside“
- Press service/Download
-
News Archiv
- Archive News 2026
- Archive News 2025
- Archive News 2024
- Archive News 2023
- Archive News 2022
- Archive News 2021
- Archive News 2020
- Archive News 2019
- Archive News 2018
- Archive News 2017
- Archive News 2016
- Archive News 2015
- Archive News 2014
- Archive News 2013
- Archive News 2012
- Archive News 2011
- Archive News 2010
- Archive News 2009
- Archive News 2008







